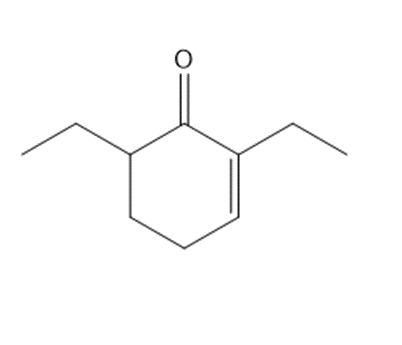
Chapter 23: Q5. (page 924)
Question: Which C-Hbonds in the following molecules are acidic because the resulting conjugate base is resonance stabilized?
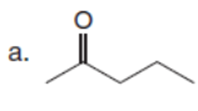
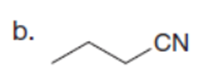
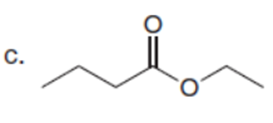
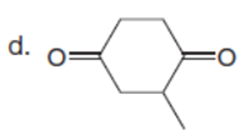
Short Answer
Answer
The most acidic C-H bonds are shown below.
Learning Materials
Features
Discover
Chapter 23: Q5. (page 924)
Question: Which C-Hbonds in the following molecules are acidic because the resulting conjugate base is resonance stabilized?




Answer
The most acidic C-H bonds are shown below.
All the tools & learning materials you need for study success - in one app.
Get started for free
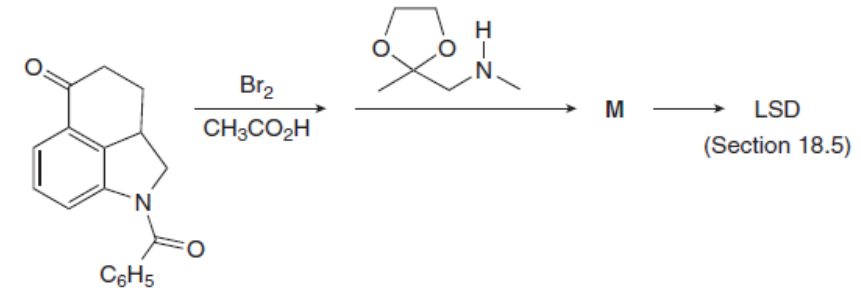
Question: Identify the product M of the following two-step reaction sequence. M was converted to the hallucinogen LSD (Figure 18.4) in several steps.
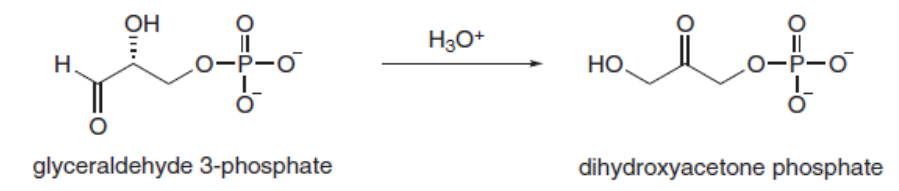
Question: During the metabolism of glucose, glyceraldehyde 3-phosphate is converted to dihydroxyacetone phosphate by a process that involves two keto-enol tautomerizations. Draw a stepwise mechanism for this reaction in the presence of acid.
Question: Synthesize each compound from diethyl malonate. You may use any other organic or inorganic reagents.
Question:What cyclic product is formed from each dihalide using the malonic ester synthesis?
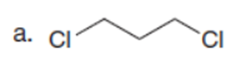
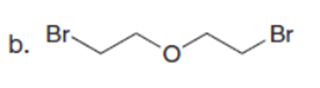
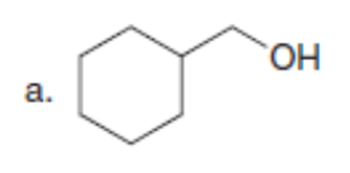
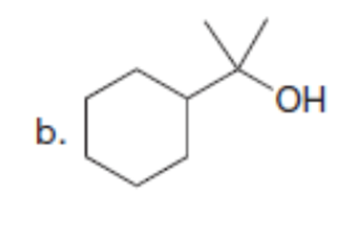
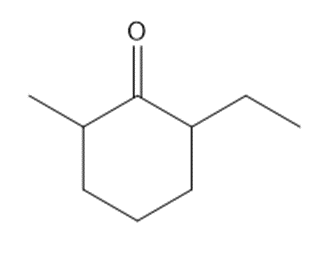
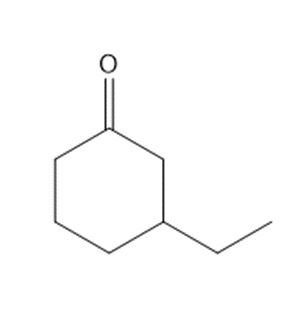
Question: Synthesize each compound from cyclohexanone and organic halides having 4 C’s. You may use any other inorganic reagents.
a.
b.
c.
What do you think about this solution?
We value your feedback to improve our textbook solutions.