Chapter 23: Q63. (page 924)
Question: Synthesize each compound from cyclohexanone and organic halides having 4 C’s. You may use any other inorganic reagents.
a.
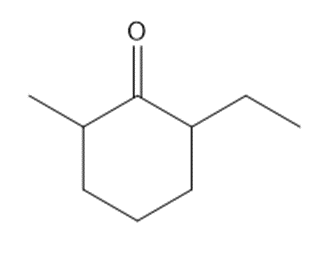
b.
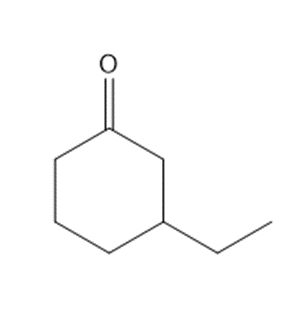
c.
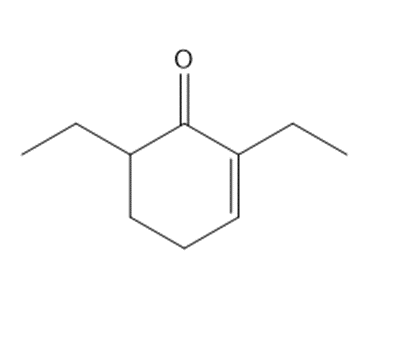
Short Answer
Answer
a.
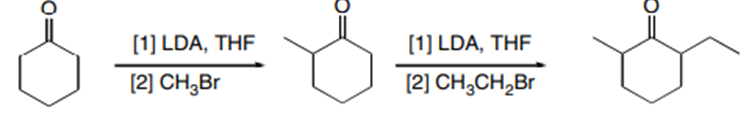
b.
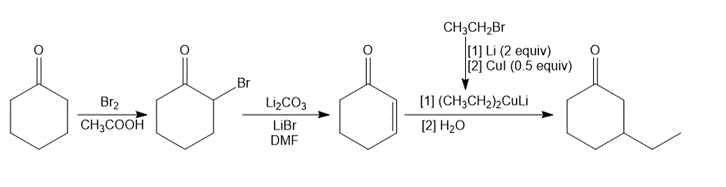
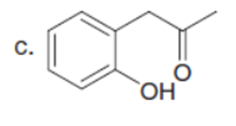
c.

Learning Materials
Features
Discover
Chapter 23: Q63. (page 924)
Question: Synthesize each compound from cyclohexanone and organic halides having 4 C’s. You may use any other inorganic reagents.
a.

b.

c.

Answer
a.

b.

c.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Devise a stepwise mechanism for the following reaction.
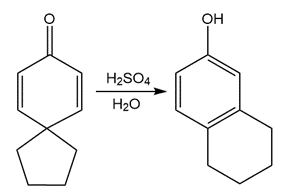
Question: Treatment of α, β-unsaturated carbonyl compound X with base forms the diastereomer Y. Write a stepwise mechanism for this reaction. Explain why one stereogenic center changes configuration but the other does not.

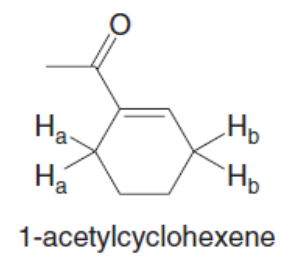
Question: Why is the pKa of the Ha protons in 1-acetylcyclohexene higher than the pKaof the Hbprotons?
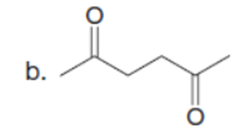
Question: What hydrogen atoms in each compound have a pka≤ 25?

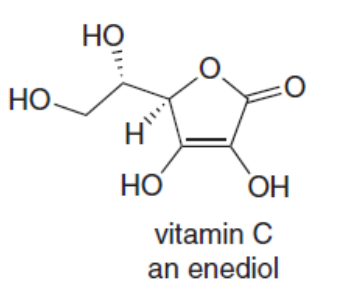
Question: Vitamin C is a stable enediol. Draw the structure of the two keto tautomers in equilibrium with the enediol and explain why the enediol is more stable than the other tautomer.
What do you think about this solution?
We value your feedback to improve our textbook solutions.