Chapter 23: Q34. (page 924)
Question: What hydrogen atoms in each compound have a pka≤ 25?
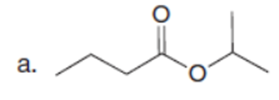

Short Answer
Answer
The hydrogen atoms in each compound have a pKa ≤ 25, as shown below:
Learning Materials
Features
Discover
Chapter 23: Q34. (page 924)
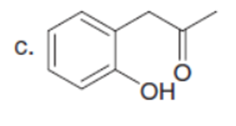
Question: What hydrogen atoms in each compound have a pka≤ 25?



Answer
The hydrogen atoms in each compound have a pKa ≤ 25, as shown below:
All the tools & learning materials you need for study success - in one app.
Get started for free
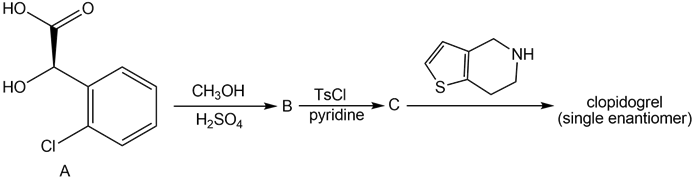
Question: Clopidogrel is the generic name for Plavix, a drug used to prevent the formation of blood clots in patients that have a history of heart attacks or strokes. A single enantiomer of clopidogrel can be prepared in three steps from the chiral α-hydroxy acid A. Identify B and C in the following reaction sequence, and designate the configuration of the enantiomer formed by this route as R or S
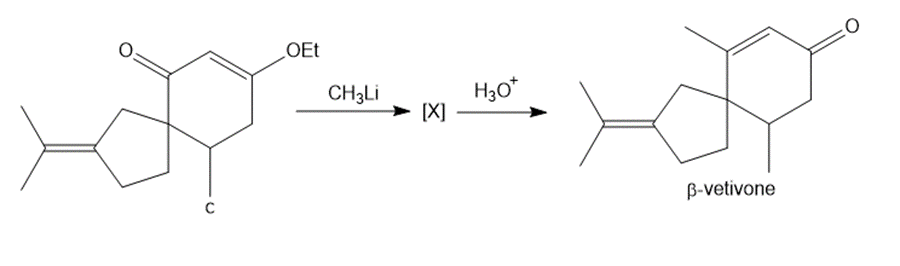
Question: The last step in the synthesis of β-vetivone (Problem 23.61) involves treatment of C with CH3Li to form an intermediate X, which forms β-vetivone with aqueous acid. Identify the structure of X and draw a mechanism for converting X to β-vetivone.
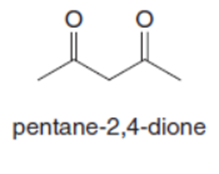
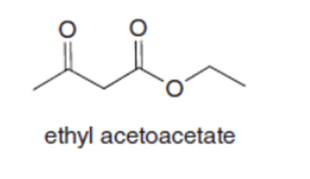
Question: Both pentane-2,4-dione and ethyl acetoacetate have two carbonyl groups separated by a single carbon atom. Although an equilibrium mixture of pentane-2,4-dione tautomers contains 76% of the enol forms, an equilibrium mixture of ethyl acetoacetate tautomers contains only 8% of the enol forms. Suggest a reason for this difference.
Question:Explain each observation: (a) When (R)-2-methylcyclohexanone is treated with NaOH in H2O , the optically active solution gradually loses optical activity. (b) When (R)-3-methylcyclohexanone is treated with NaOH in H20 , the solution remains optically active.
Question:What alkyl halides are needed to prepare each ketone using the acetoacetic ester synthesis?
What do you think about this solution?
We value your feedback to improve our textbook solutions.