Chapter 23: Q35. (page 924)
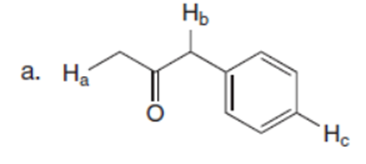
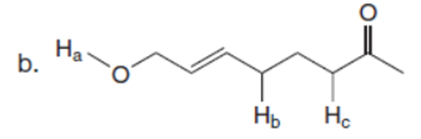
Question: Rank the labeled protons in each compound in order of increasing acidity.
Short Answer
Answer
The order of acidity of the labeled protons in each compound is shown below:
Learning Materials
Features
Discover
Chapter 23: Q35. (page 924)
Question: Rank the labeled protons in each compound in order of increasing acidity.


Answer
The order of acidity of the labeled protons in each compound is shown below:
All the tools & learning materials you need for study success - in one app.
Get started for free
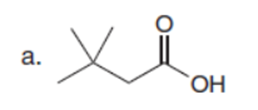
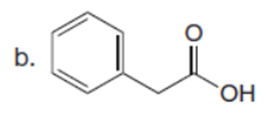
Question:Explain why each of the following carboxylic acids cannot be prepared by a malonic ester synthesis.
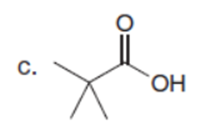
Question:What alkyl halides are needed to prepare each ketone using the acetoacetic ester synthesis?
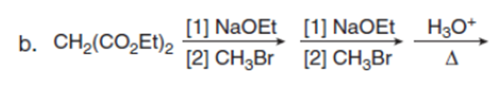
Question:Draw the product of each reaction.

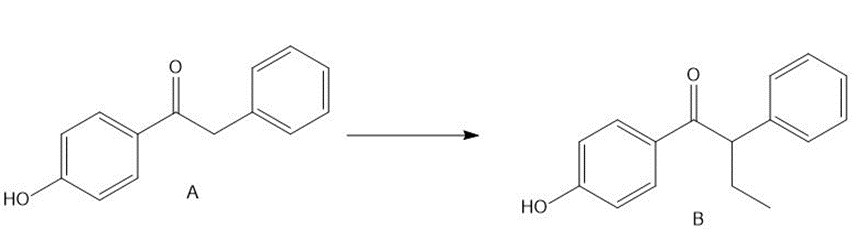
Question: Treatment of ketone A with LDA followed by CH3CH2 did not form the desired alkylation product B. What product was formed instead? Devise a multistep method to convert A to B, a synthetic intermediate used to prepare the anticancer drug tamoxifen (Section 23.8C and the chapter-opening molecule).
Question: What product is formed when each compound is treated first with LDA in THF solution at low temperature, followed by CH3CH2l?
What do you think about this solution?
We value your feedback to improve our textbook solutions.