Chapter 23: Q36. (page 924)
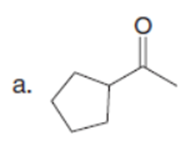
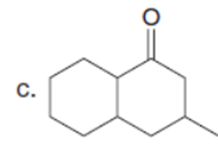
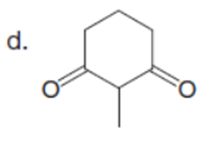
Question:What is the major enolate (or carbanion) formed when each compound is treated with LDA?

Short Answer
Answer
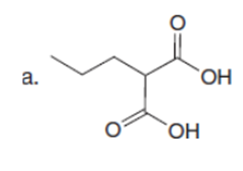
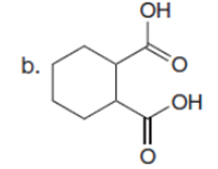
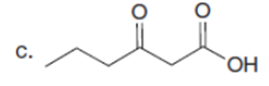
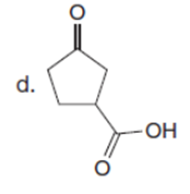
The major enolate (or carbanion) formed when each compound is treated with LDA is shown below:-
Learning Materials
Features
Discover
Chapter 23: Q36. (page 924)
Question:What is the major enolate (or carbanion) formed when each compound is treated with LDA?




Answer
The major enolate (or carbanion) formed when each compound is treated with LDA is shown below:-
All the tools & learning materials you need for study success - in one app.
Get started for free
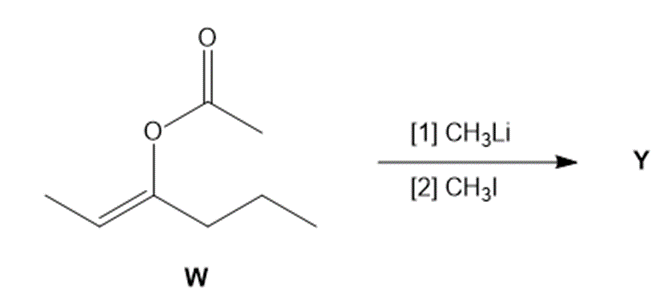
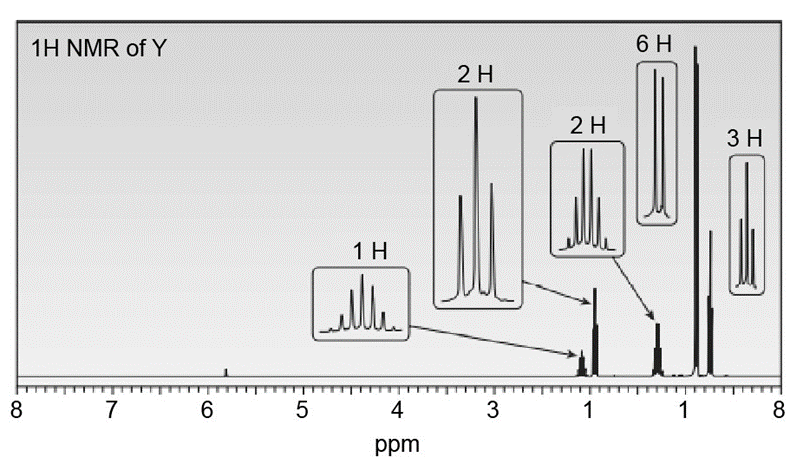
Question: Treatment of W with CH3Li, followed by CH3l, affords compound Y (C7H14O ) as the major product. Y shows a strong absorption in its IR spectrum at , and its 1 H NMR spectrum is given below. (a) Propose a structure for Y. (b) Draw a stepwise mechanism for the conversion of W to Y.
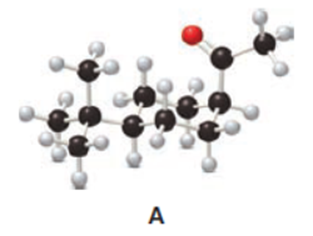
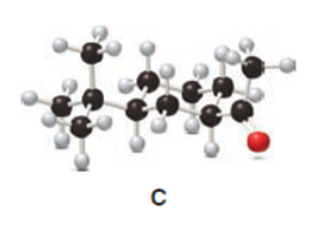
Question: The cis ketone A is isomerized to a trans ketone B with aqueous NaOH. A similar isomerization does not occur with ketone C. (a) Draw the structure of B using a chair cyclohexane. (b) Label the substituents in C as cis or trans, and explain the difference in reactivity.
Question: Draw enol tautomer(s) for each compound. Ignore stereoisomers.


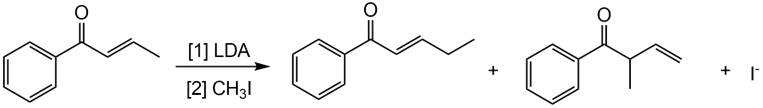
Question: Draw a stepwise mechanism showing how two alkylation products are formed in the following reaction.
Question:Which of the following compounds will readily lose CO2when heated?
What do you think about this solution?
We value your feedback to improve our textbook solutions.