Chapter 23: Q29. (page 924)
Question: Draw enol tautomer(s) for each compound. Ignore stereoisomers.
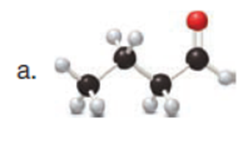
Short Answer
Answer
The enol tautomer(s) for each compound is shown below:
Learning Materials
Features
Discover
Chapter 23: Q29. (page 924)
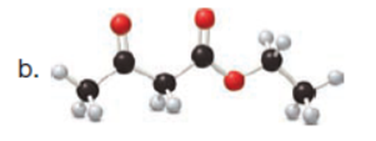
Question: Draw enol tautomer(s) for each compound. Ignore stereoisomers.


Answer
The enol tautomer(s) for each compound is shown below:
All the tools & learning materials you need for study success - in one app.
Get started for free
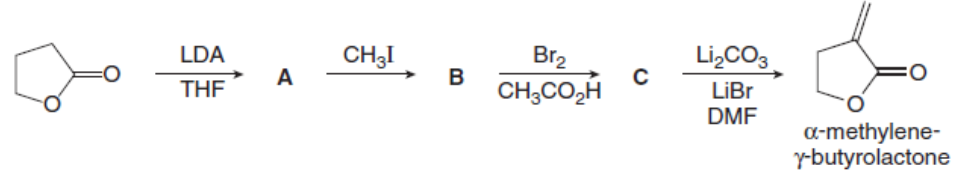
Question: Identify A, B, and C, intermediates in the synthesis of the five-membered ring called an -methylene- -butyrolactone. This heterocyclic ring system is present in some antitumor agents.
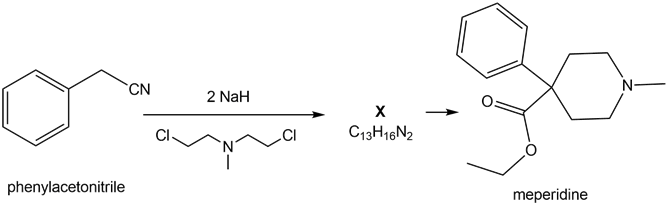
Question: A key step in the synthesis of the narcotic analgesic meperidine (trade name Demerol) is the conversion of phenyl acetonitrile to X. (a) What is the structure of X? (b) What reactions convert X to meperidine?
Question:Which of the following compounds will readily lose CO2when heated?
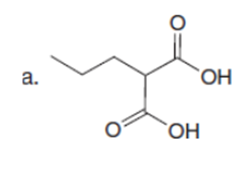
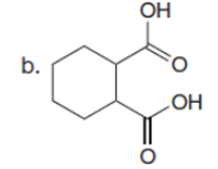
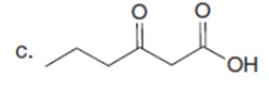
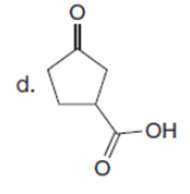
Question: Draw a stepwise mechanism for the following reaction.
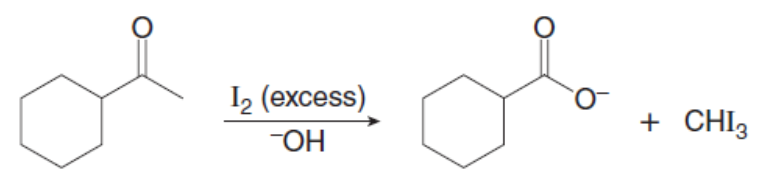
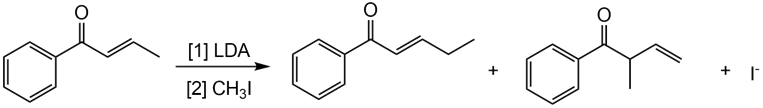
Question: Draw a stepwise mechanism showing how two alkylation products are formed in the following reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.