Chapter 23: Q42. (page 924)
Question: Draw a stepwise mechanism for the following reaction.
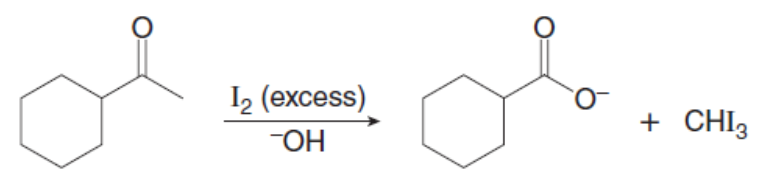
Short Answer
Answer
The stepwise mechanism of the haloform reaction is shown below.
Learning Materials
Features
Discover
Chapter 23: Q42. (page 924)
Question: Draw a stepwise mechanism for the following reaction.

Answer
The stepwise mechanism of the haloform reaction is shown below.
All the tools & learning materials you need for study success - in one app.
Get started for free
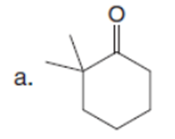
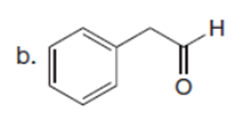
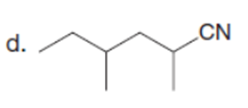
Question: Draw the product formed when each starting material is treated with LDA in THF solution at –780C.

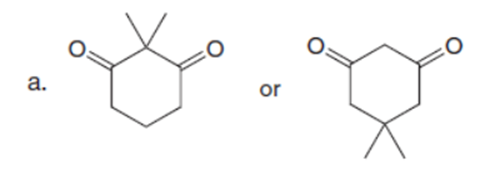
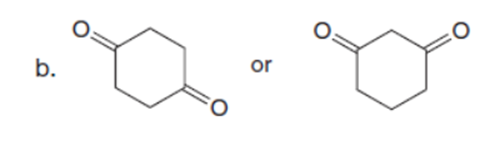
Question: Which carbonyl compound in each pair exhibits the higher percentage of the enol tautomer?
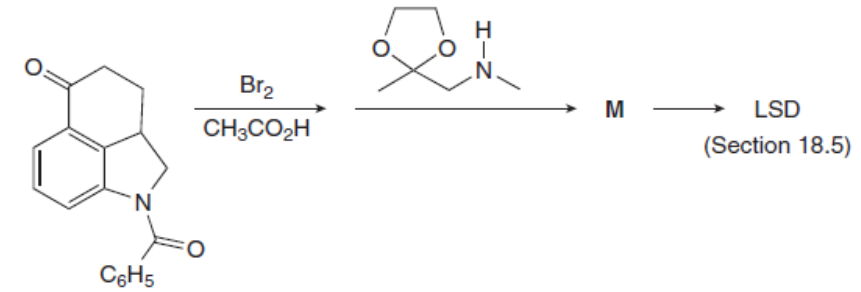
Question: Identify the product M of the following two-step reaction sequence. M was converted to the hallucinogen LSD (Figure 18.4) in several steps.
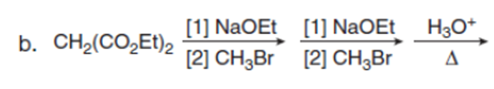
Question:Draw the product of each reaction.

Question: (–)-Hyoscyamine, an optically active drug used to treat gastrointestinal disorders, is isolated from Atropa belladonna, the deadly nightshade plant, by a basic aqueous extraction procedure. If too much base is used during isolation, optically inactive material is isolated. (a) Explain this result by drawing a stepwise mechanism. (b) Explain why littorine, an isomer isolated from the tailflower plant in Australia, can be obtained optically pure regardless of the amount of base used during isolation.
What do you think about this solution?
We value your feedback to improve our textbook solutions.