Chapter 23: Q7. (page 924)
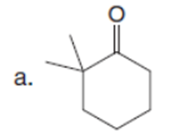
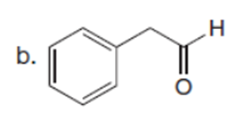
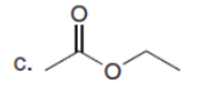
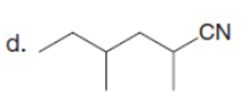
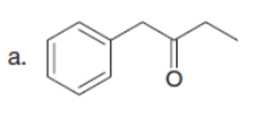
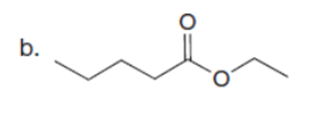
Question: Draw the product formed when each starting material is treated with LDA in THF solution at –780C.
Short Answer
Answer
The products formed when each starting material is treated with LDA are shown below.
Learning Materials
Features
Discover
Chapter 23: Q7. (page 924)
Question: Draw the product formed when each starting material is treated with LDA in THF solution at –780C.




Answer
The products formed when each starting material is treated with LDA are shown below.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Capsaicin, the spicy component of hot peppers, can be prepared from amine X and acid chloride Y. Devise a synthesis of Y from (E)-6-methylhept-4-en-1-ol [(CH3)2 CHCH=CH(CH2)3OH],CH2(CO2Et)2 , and any required inorganic reagents.
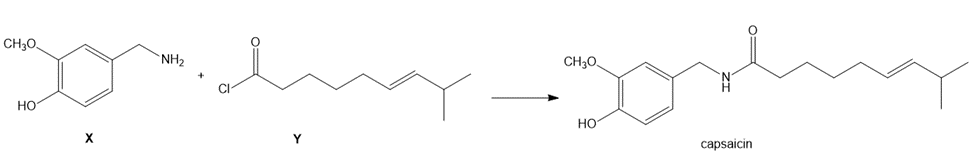
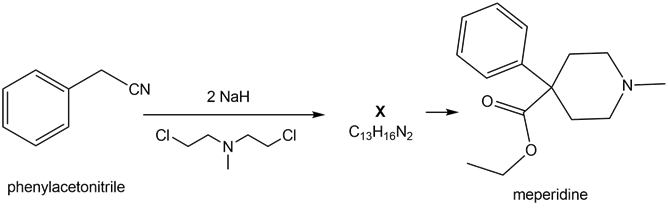
Question: A key step in the synthesis of the narcotic analgesic meperidine (trade name Demerol) is the conversion of phenyl acetonitrile to X. (a) What is the structure of X? (b) What reactions convert X to meperidine?
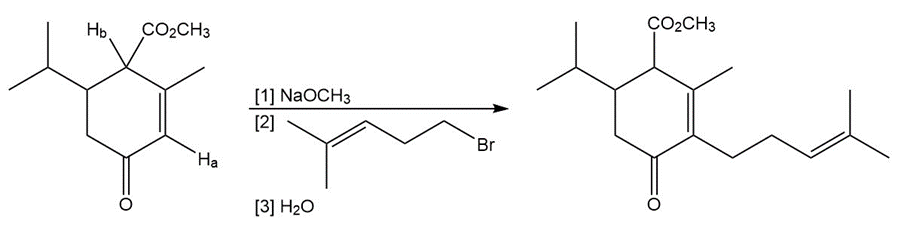
Question: Explain why Ha is much less acidic than Hb . Then draw a mechanism for the following reaction.
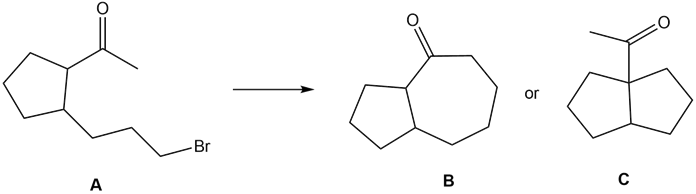
Question: What reaction conditions—base, solvent, and temperature—are needed to convert ketone A to either B or C by an intramolecular alkylation reaction?
Question: Draw enol tautomer(s) for each compound.

What do you think about this solution?
We value your feedback to improve our textbook solutions.