Chapter 23: Q31. (page 924)
Question: Draw enol tautomer(s) for each compound.
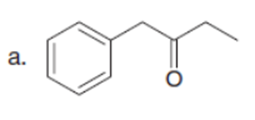

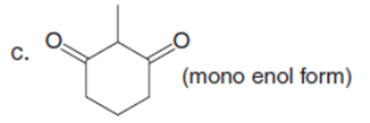
Short Answer
Answer
The enol tautomer(s) for each compound is shown below:
Learning Materials
Features
Discover
Chapter 23: Q31. (page 924)
Question: Draw enol tautomer(s) for each compound.



Answer
The enol tautomer(s) for each compound is shown below:
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: When phenylacetaldehyde (C6H5CH2CHO) is dissolved in with added DCl, the hydrogen atomsαto the carbonyl are gradually replaced by deuterium atoms. Write a mechanism for this process that involves enols as intermediates.
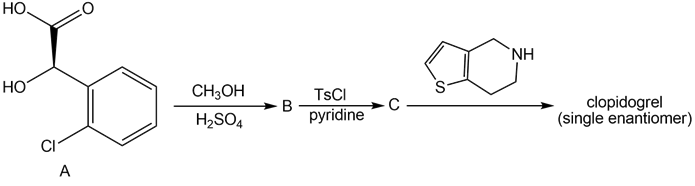
Question: Clopidogrel is the generic name for Plavix, a drug used to prevent the formation of blood clots in patients that have a history of heart attacks or strokes. A single enantiomer of clopidogrel can be prepared in three steps from the chiral α-hydroxy acid A. Identify B and C in the following reaction sequence, and designate the configuration of the enantiomer formed by this route as R or S
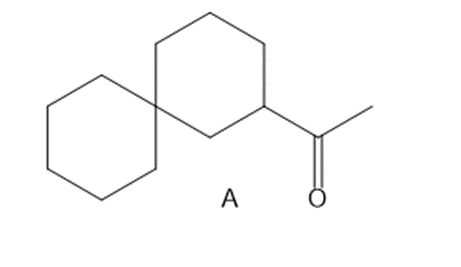
Question:
(a) Draw two different haloketones that can form A by an intramolecular alkylation reaction.
(b) How can A be synthesized by an acetoacetic ester synthesis?
Question: What enolate is formed when each ketone is treated with LDA in THF solution? What enolate is formed when these same ketones are treated with NaOCH3in CH3OHsolution?



Question: Use the malonic ester synthesis to prepare each carboxylic acid.
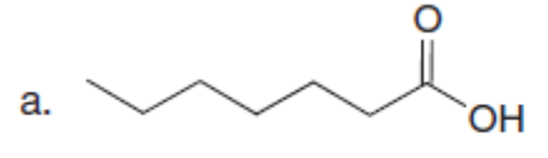
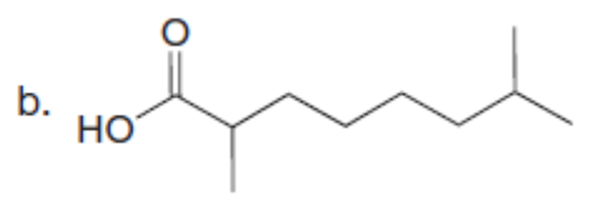
What do you think about this solution?
We value your feedback to improve our textbook solutions.