Chapter 23: Q44. (page 924)
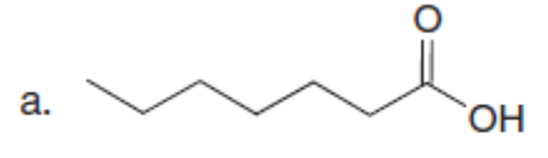
Question: Use the malonic ester synthesis to prepare each carboxylic acid.

Short Answer
Answer
The starting materials needed to prepare each carboxylic acid by the malonic ester synthesis are shown below.
Learning Materials
Features
Discover
Chapter 23: Q44. (page 924)
Question: Use the malonic ester synthesis to prepare each carboxylic acid.


Answer
The starting materials needed to prepare each carboxylic acid by the malonic ester synthesis are shown below.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question:Bupropion, sold under the trade name of Zyban, is an antidepressant that was approved to aid smoking cessation in 1997. Devise a synthesis of bupropion from benzene, organic compounds that have fewer than five carbons, and any required inorganic reagents.

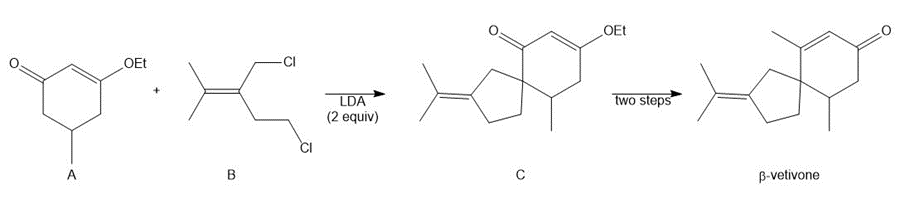
Question: A key step in the synthesis of β-vetivone, a major constituent of vetiver, a perennial grass found in tropical and subtropical regions of the world, involved the reaction of compound A and dihalide B with two equivalents of LDA to form C. Draw a stepwise mechanism for this reaction. β-Vetivone contains a spiro ring system—that is, two rings that share a single carbon atom.
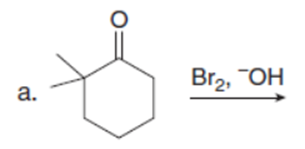
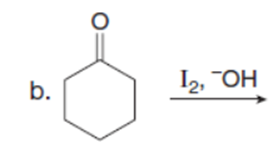
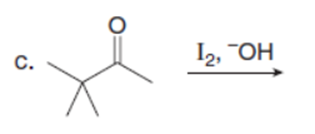
Question: Draw the products of each reaction. Assume excess halogen is present.
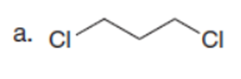
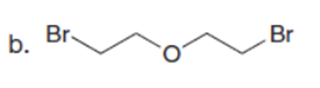
Question:What cyclic product is formed from each dihalide using the malonic ester synthesis?
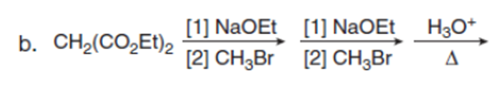
Question:Draw the product of each reaction.

What do you think about this solution?
We value your feedback to improve our textbook solutions.