Chapter 23: Q21. (page 924)
Question:Draw the product of each reaction.
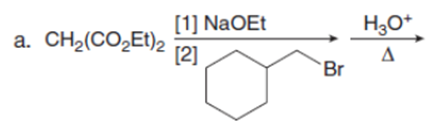
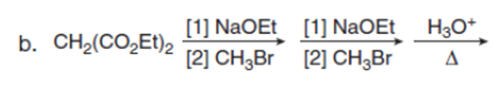
Short Answer
Answers
The product formed from each reaction are shown below:-
Learning Materials
Features
Discover
Chapter 23: Q21. (page 924)
Question:Draw the product of each reaction.


Answers
The product formed from each reaction are shown below:-
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Use the malonic ester synthesis to prepare each carboxylic acid.
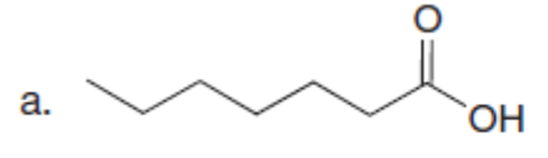
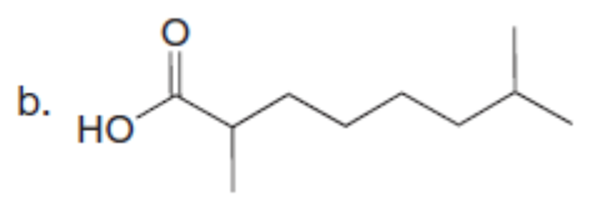
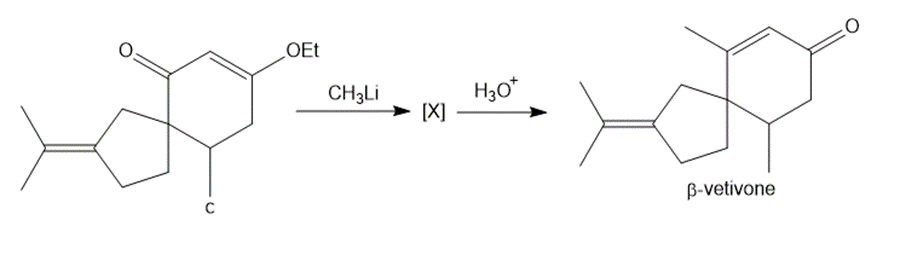
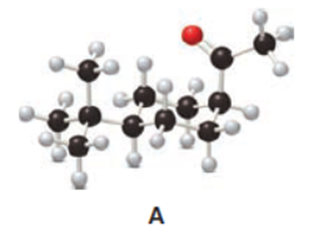
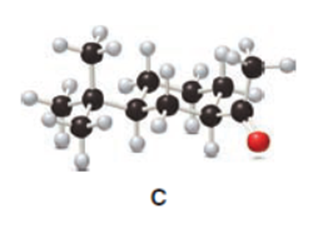
Question: The last step in the synthesis of β-vetivone (Problem 23.61) involves treatment of C with CH3Li to form an intermediate X, which forms β-vetivone with aqueous acid. Identify the structure of X and draw a mechanism for converting X to β-vetivone.
Question: Draw the products obtained (including stereochemistry) when each compound is treated with LDA, followed by CH3l.
Question: The cis ketone A is isomerized to a trans ketone B with aqueous NaOH. A similar isomerization does not occur with ketone C. (a) Draw the structure of B using a chair cyclohexane. (b) Label the substituents in C as cis or trans, and explain the difference in reactivity.
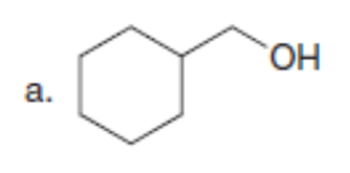
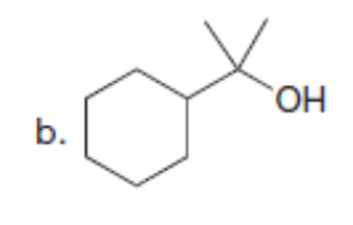
Question: Synthesize each compound from diethyl malonate. You may use any other organic or inorganic reagents.
What do you think about this solution?
We value your feedback to improve our textbook solutions.