Chapter 23: Q62. (page 924)
Question:
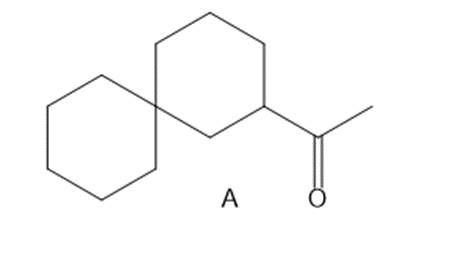
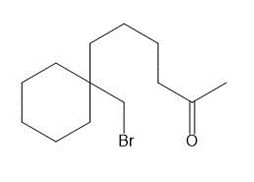
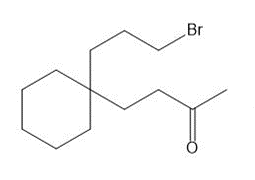
(a) Draw two different haloketones that can form A by an intramolecular alkylation reaction.
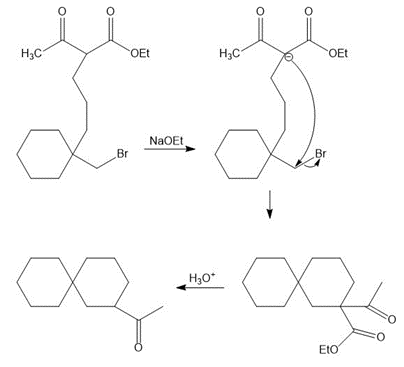
(b) How can A be synthesized by an acetoacetic ester synthesis?
Short Answer
Answer
(a)
(b)
Learning Materials
Features
Discover
Chapter 23: Q62. (page 924)
Question:
(a) Draw two different haloketones that can form A by an intramolecular alkylation reaction.
(b) How can A be synthesized by an acetoacetic ester synthesis?

Answer
(a)


(b)

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: How can pentan-2-one be converted into each compound?
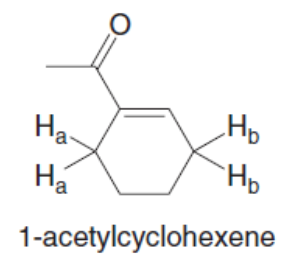
Question: Why is the pKa of the Ha protons in 1-acetylcyclohexene higher than the pKaof the Hbprotons?
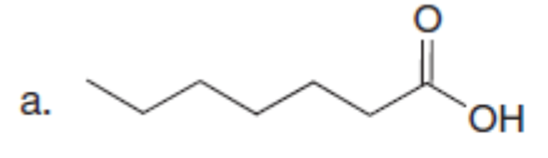
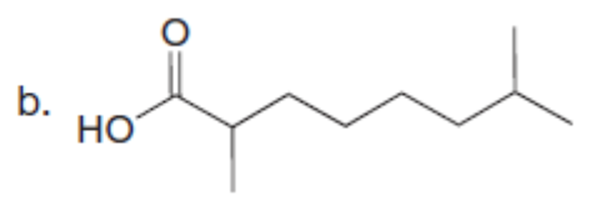
Question: Use the malonic ester synthesis to prepare each carboxylic acid.
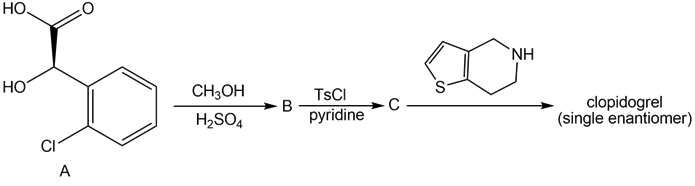
Question: Clopidogrel is the generic name for Plavix, a drug used to prevent the formation of blood clots in patients that have a history of heart attacks or strokes. A single enantiomer of clopidogrel can be prepared in three steps from the chiral α-hydroxy acid A. Identify B and C in the following reaction sequence, and designate the configuration of the enantiomer formed by this route as R or S
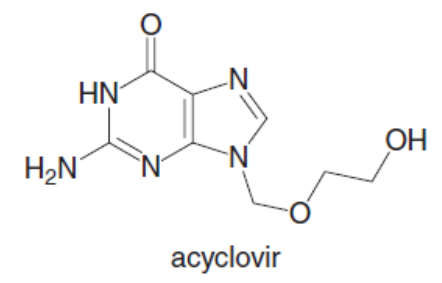
Question: Acyclovir is an effective antiviral agent used to treat the herpes simplex virus. (a) Draw the enol form of acyclovir, and explain why it is aromatic. (b) Why is acyclovir typically drawn in its keto form, despite the fact that its enol is aromatic?
What do you think about this solution?
We value your feedback to improve our textbook solutions.