Chapter 23: Q18. (page 924)
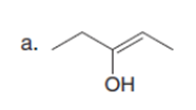
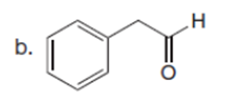
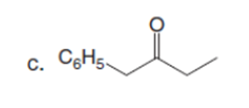
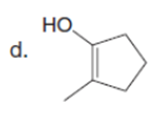
Question: How can pentan-2-one be converted into each compound?
Short Answer
Answers
The pentan-2-one be converted into each compound by the following reactions:
Learning Materials
Features
Discover
Chapter 23: Q18. (page 924)
Question: How can pentan-2-one be converted into each compound?
Answers
The pentan-2-one be converted into each compound by the following reactions:
All the tools & learning materials you need for study success - in one app.
Get started for free
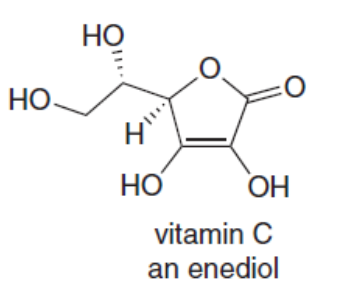
Question: Vitamin C is a stable enediol. Draw the structure of the two keto tautomers in equilibrium with the enediol and explain why the enediol is more stable than the other tautomer.
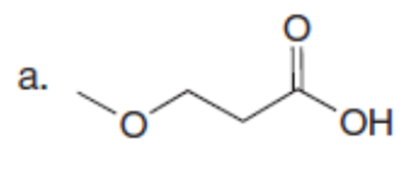
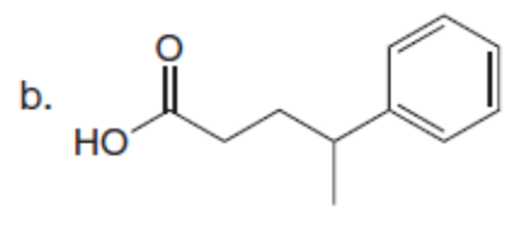
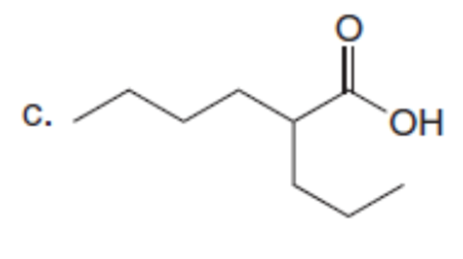
Question: What alkyl halides are needed to prepare each carboxylic acid using the malonic ester synthesis?
Question: Draw the products of each reaction. Assume excess halogen is present.
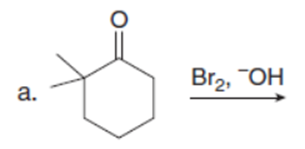
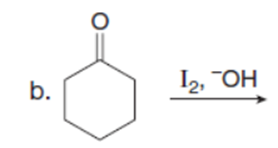
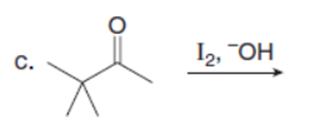
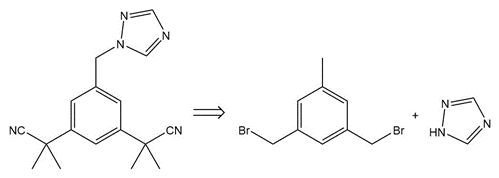
Question:Devise a synthesis of anastrozole, a drug used to reduce the recurrence of breast cancer (Section 22.18), from the given compounds. You may use any other needed organic compounds or inorganic reagents.
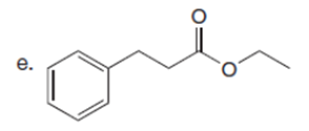
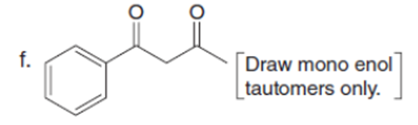
Question: Draw the enol or keto tautomer(s) of each compound.
What do you think about this solution?
We value your feedback to improve our textbook solutions.