Chapter 23: Q1. (page 924)
Question: Draw the enol or keto tautomer(s) of each compound.
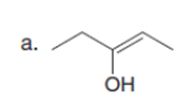
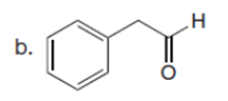
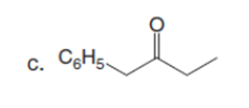
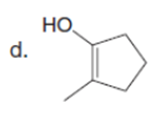
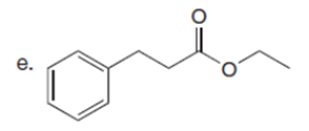
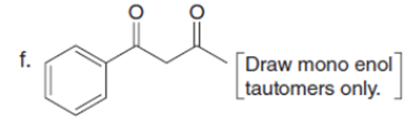
Short Answer
Answer
The enol or keto tautomer(s) of each compound are shown below.
Learning Materials
Features
Discover
Chapter 23: Q1. (page 924)
Question: Draw the enol or keto tautomer(s) of each compound.






Answer
The enol or keto tautomer(s) of each compound are shown below.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: A key step in the synthesis of the narcotic analgesic meperidine (trade name Demerol) is the conversion of phenyl acetonitrile to X. (a) What is the structure of X? (b) What reactions convert X to meperidine?
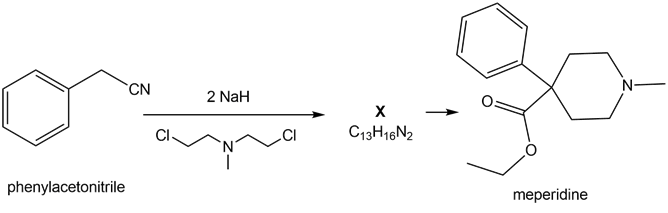
Question:The enolate derived from diethyl malonate reacts with a variety of electrophiles (not just alkyl halides) to form new carbon-carbon bonds. With this in mind, draw the products formed when Na+ –CH(CO2Et)2 reacts with each electrophile, followed by treatment with H2O.
Question:What alkyl halides are needed to prepare each ketone using the acetoacetic ester synthesis?
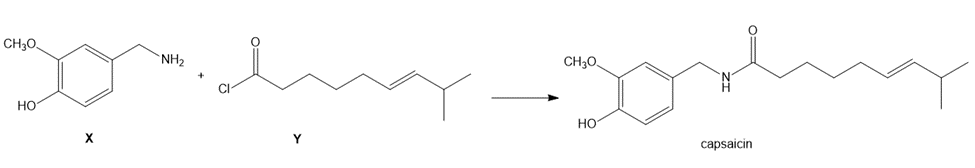
Question: Capsaicin, the spicy component of hot peppers, can be prepared from amine X and acid chloride Y. Devise a synthesis of Y from (E)-6-methylhept-4-en-1-ol [(CH3)2 CHCH=CH(CH2)3OH],CH2(CO2Et)2 , and any required inorganic reagents.
Question: As we learned in Chapter 20, organolithium reagents (RLi) are strong bases that readily react with acidic protons. Why aren’t organolithium reagents used to generate enolates?
What do you think about this solution?
We value your feedback to improve our textbook solutions.