Chapter 23: Q70. (page 924)
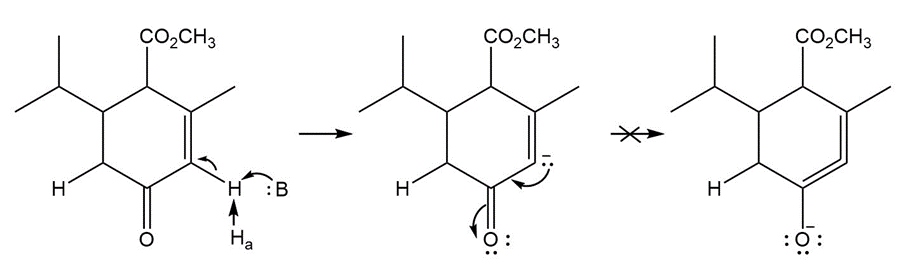
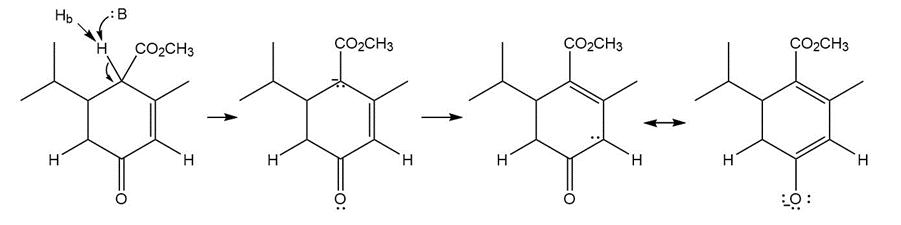
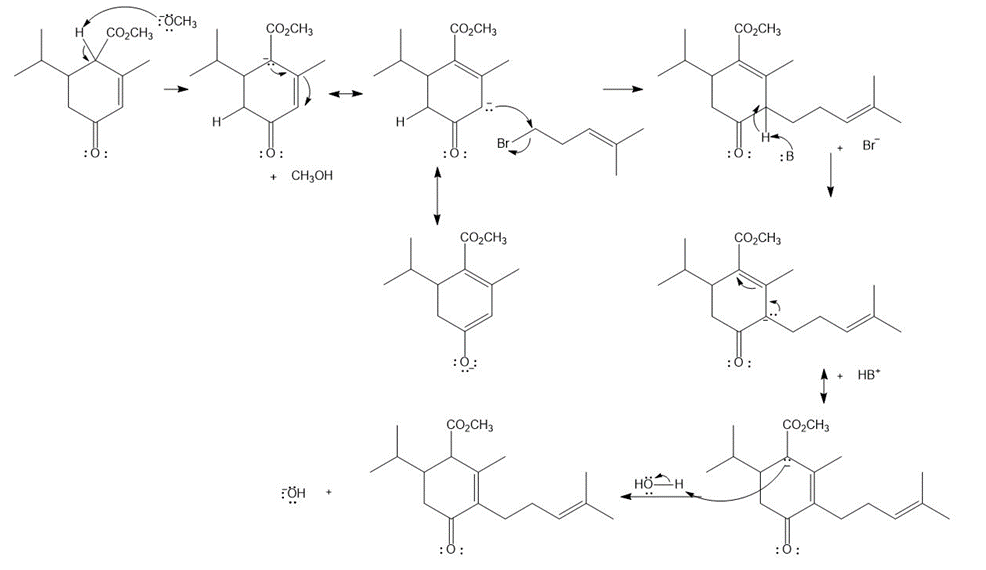
Question: Explain why Ha is much less acidic than Hb . Then draw a mechanism for the following reaction.
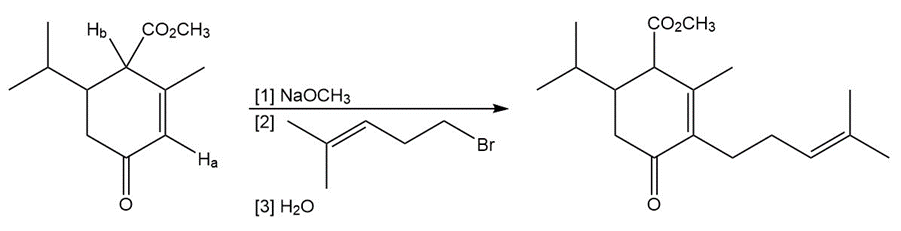
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 23: Q70. (page 924)
Question: Explain why Ha is much less acidic than Hb . Then draw a mechanism for the following reaction.

Answer



All the tools & learning materials you need for study success - in one app.
Get started for free
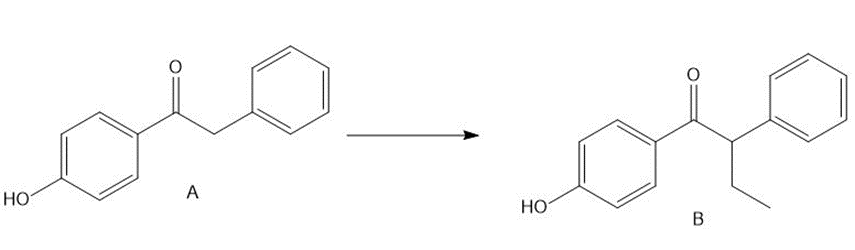
Question: Treatment of ketone A with LDA followed by CH3CH2 did not form the desired alkylation product B. What product was formed instead? Devise a multistep method to convert A to B, a synthetic intermediate used to prepare the anticancer drug tamoxifen (Section 23.8C and the chapter-opening molecule).
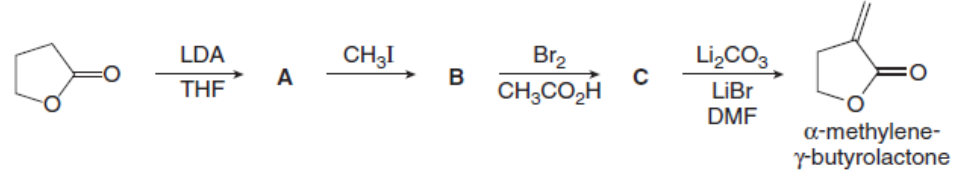
Question: Identify A, B, and C, intermediates in the synthesis of the five-membered ring called an -methylene- -butyrolactone. This heterocyclic ring system is present in some antitumor agents.
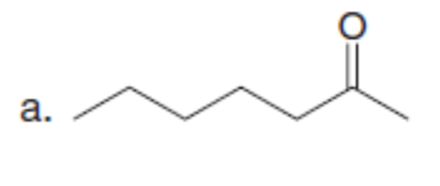
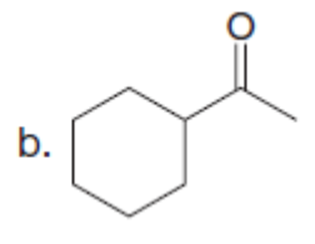
Question: What enolate is formed when each ketone is treated with LDA in THF solution? What enolate is formed when these same ketones are treated with NaOCH3in CH3OHsolution?
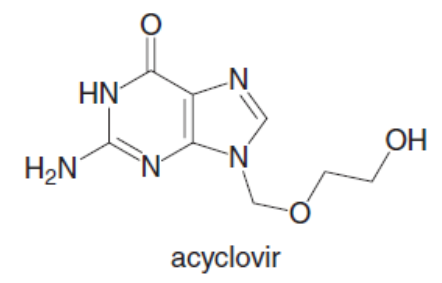
Question: Acyclovir is an effective antiviral agent used to treat the herpes simplex virus. (a) Draw the enol form of acyclovir, and explain why it is aromatic. (b) Why is acyclovir typically drawn in its keto form, despite the fact that its enol is aromatic?
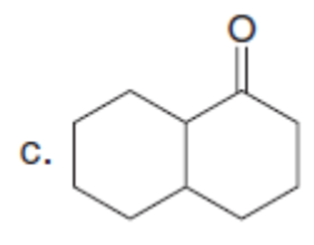
Question:The enolate derived from diethyl malonate reacts with a variety of electrophiles (not just alkyl halides) to form new carbon-carbon bonds. With this in mind, draw the products formed when Na+ –CH(CO2Et)2 reacts with each electrophile, followed by treatment with H2O.
What do you think about this solution?
We value your feedback to improve our textbook solutions.