Chapter 23: Q57. (page 924)
Question: Draw a stepwise mechanism showing how two alkylation products are formed in the following reaction.
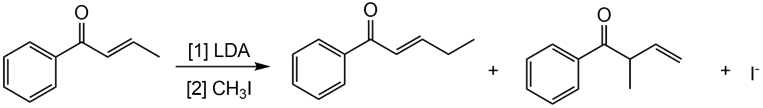
Short Answer
Answer
Stepwise mechanism:
Step 1
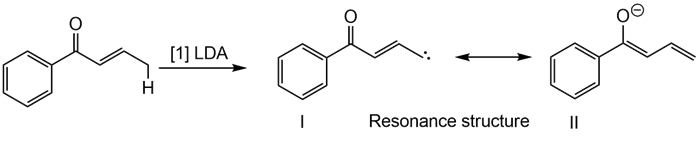
Step 2
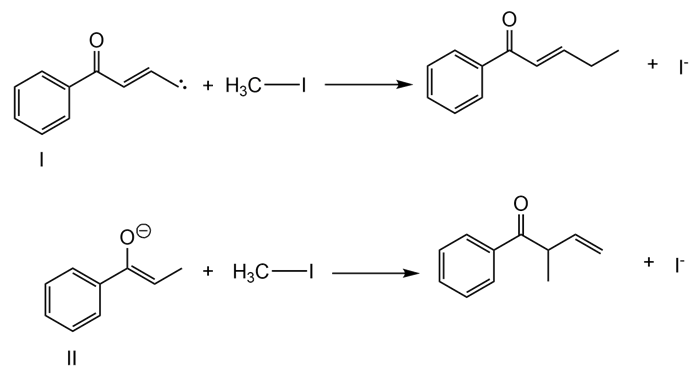
Learning Materials
Features
Discover
Chapter 23: Q57. (page 924)
Question: Draw a stepwise mechanism showing how two alkylation products are formed in the following reaction.

Answer
Stepwise mechanism:
Step 1

Step 2

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: As we learned in Chapter 20, organolithium reagents (RLi) are strong bases that readily react with acidic protons. Why aren’t organolithium reagents used to generate enolates?
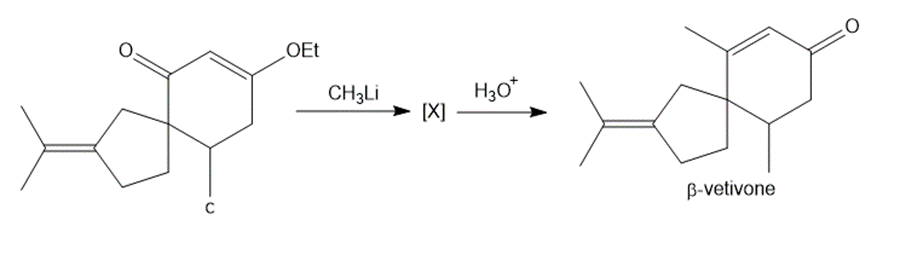
Question: The last step in the synthesis of β-vetivone (Problem 23.61) involves treatment of C with CH3Li to form an intermediate X, which forms β-vetivone with aqueous acid. Identify the structure of X and draw a mechanism for converting X to β-vetivone.
Question: Draw the product formed when each starting material is treated with LDA in THF solution at –780C.
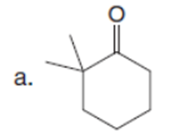
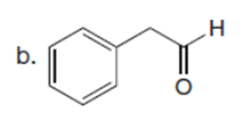
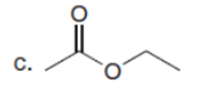
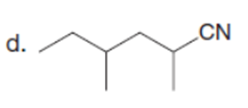
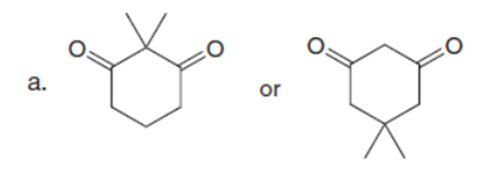
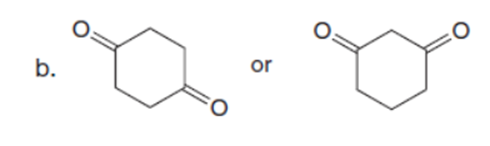
Question: Which carbonyl compound in each pair exhibits the higher percentage of the enol tautomer?
Question:What alkyl halides are needed to prepare each ketone using the acetoacetic ester synthesis?
What do you think about this solution?
We value your feedback to improve our textbook solutions.