Chapter 23: Q49. (page 924)
Question: Synthesize each compound from ethyl acetoacetate. You may use any other organic or inorganic reagents.
Short Answer
Answer
The compounds synthesized using the acetoacetic ester synthesis are shown below.
Learning Materials
Features
Discover
Chapter 23: Q49. (page 924)
Question: Synthesize each compound from ethyl acetoacetate. You may use any other organic or inorganic reagents.
Answer
The compounds synthesized using the acetoacetic ester synthesis are shown below.
All the tools & learning materials you need for study success - in one app.
Get started for free
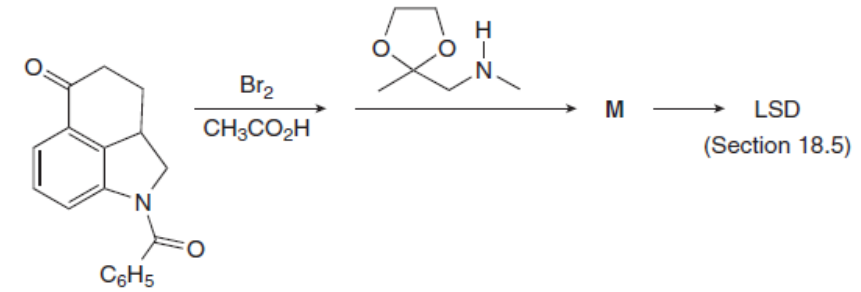
Question: Identify the product M of the following two-step reaction sequence. M was converted to the hallucinogen LSD (Figure 18.4) in several steps.
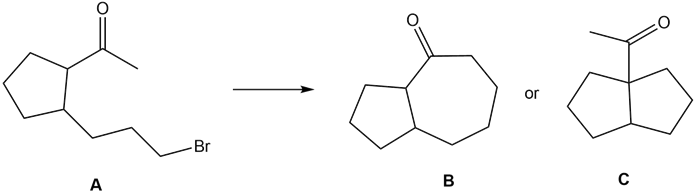
Question: What reaction conditions—base, solvent, and temperature—are needed to convert ketone A to either B or C by an intramolecular alkylation reaction?
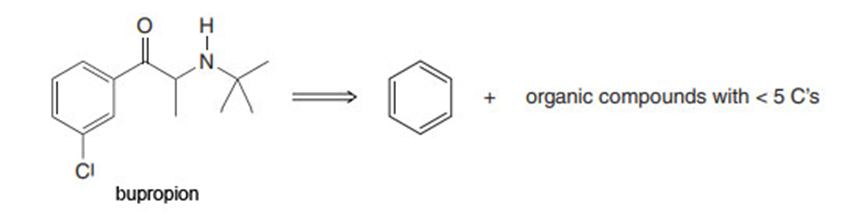
Question:Bupropion, sold under the trade name of Zyban, is an antidepressant that was approved to aid smoking cessation in 1997. Devise a synthesis of bupropion from benzene, organic compounds that have fewer than five carbons, and any required inorganic reagents.
Question: Synthesize each compound from cyclohexanone and organic halides having 4 C’s. You may use any other inorganic reagents.
a.
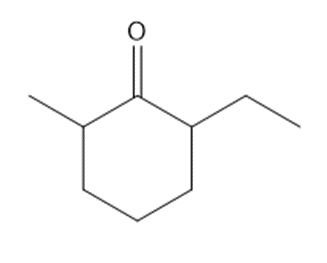
b.
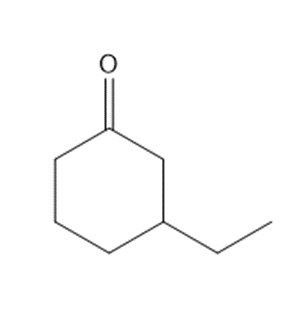
c.
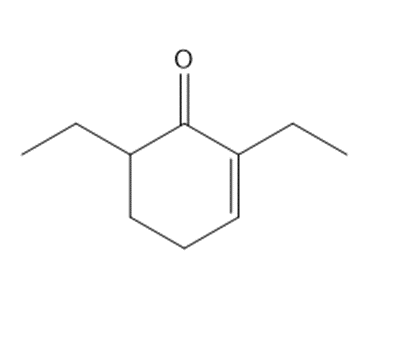
Question:The enolate derived from diethyl malonate reacts with a variety of electrophiles (not just alkyl halides) to form new carbon-carbon bonds. With this in mind, draw the products formed when Na+ –CH(CO2Et)2 reacts with each electrophile, followed by treatment with H2O.
What do you think about this solution?
We value your feedback to improve our textbook solutions.