Chapter 23: Q6. (page 924)
Question: Rank the protons in the labeled CH2groups in order of increasing acidity, and explain why you chose this order.
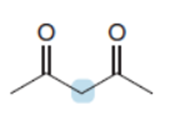
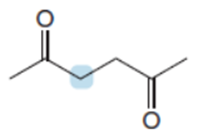
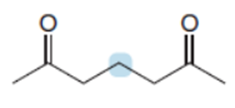
Short Answer
Answer
The order of acidity among the given compounds is shown below.
Learning Materials
Features
Discover
Chapter 23: Q6. (page 924)
Question: Rank the protons in the labeled CH2groups in order of increasing acidity, and explain why you chose this order.



Answer
The order of acidity among the given compounds is shown below.
All the tools & learning materials you need for study success - in one app.
Get started for free
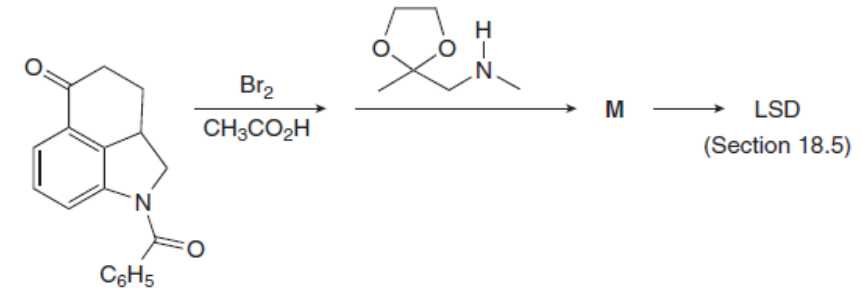
Question: Identify the product M of the following two-step reaction sequence. M was converted to the hallucinogen LSD (Figure 18.4) in several steps.
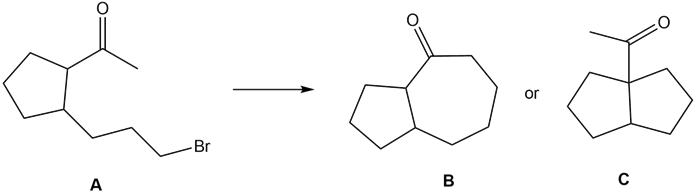
Question: What reaction conditions—base, solvent, and temperature—are needed to convert ketone A to either B or C by an intramolecular alkylation reaction?
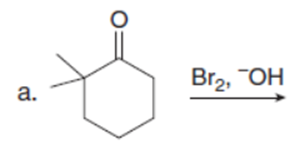
Question: Draw the products of each reaction. Assume excess halogen is present.
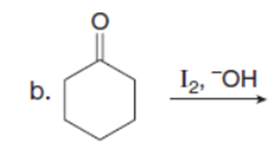
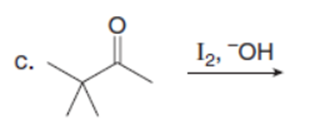
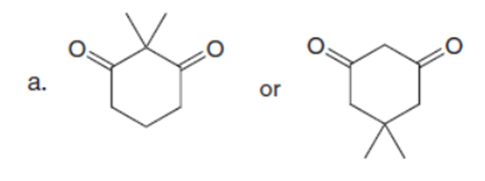
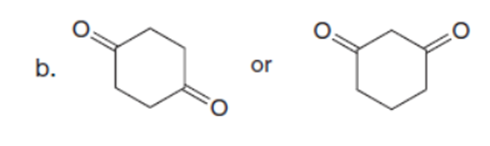
Question: Which carbonyl compound in each pair exhibits the higher percentage of the enol tautomer?
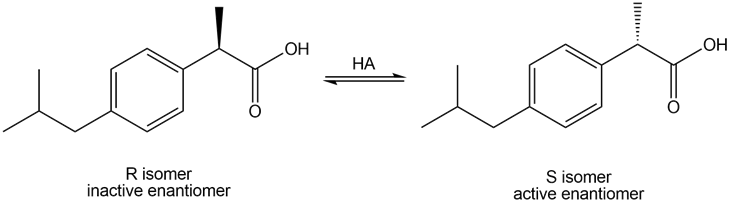
Question: Although ibuprofen is sold as a racemic mixture, only the S enantiomer acts as an analgesic. In the body, however, some of the R enantiomer is converted to the S isomer by tautomerization to an enol and then protonation to regenerate the carbonyl compound. Write a stepwise mechanism for this isomerization.
What do you think about this solution?
We value your feedback to improve our textbook solutions.