Chapter 23: Substitution Reactions of Carbonyl Compounds at the α Carbon
Q28.
Question: Nabumetone is a pain reliever and anti-inflammatory agent sold under the brand name of Relafen.
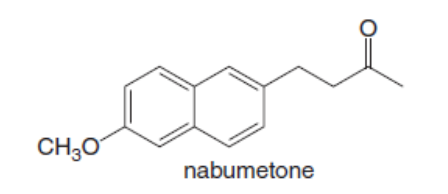
a. Write out a synthesis of nabumetone from ethyl acetoacetate.
b. What ketone and alkyl halide are needed to synthesize nabumetone by direct enolate alkylation?
Q29.
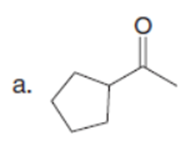
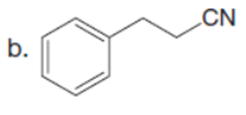
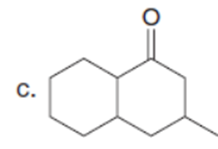
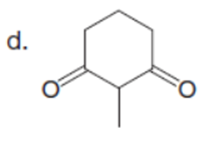
Question: Draw enol tautomer(s) for each compound. Ignore stereoisomers.
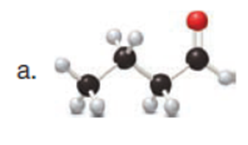
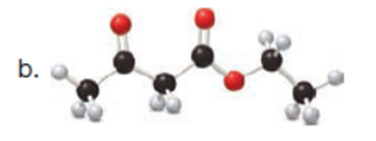
Q3.
Question: When phenylacetaldehyde (C6H5CH2CHO) is dissolved in with added DCl, the hydrogen atomsαto the carbonyl are gradually replaced by deuterium atoms. Write a mechanism for this process that involves enols as intermediates.
Q30.
Question: The cis ketone A is isomerized to a trans ketone B with aqueous NaOH. A similar isomerization does not occur with ketone C. (a) Draw the structure of B using a chair cyclohexane. (b) Label the substituents in C as cis or trans, and explain the difference in reactivity.
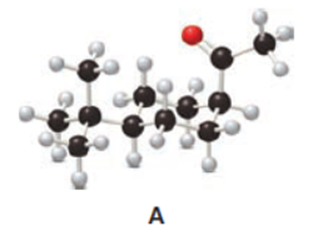
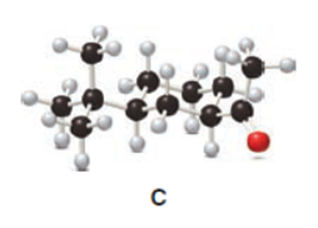
Q31.
Question: Draw enol tautomer(s) for each compound.
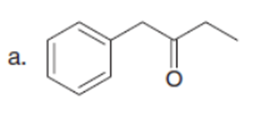
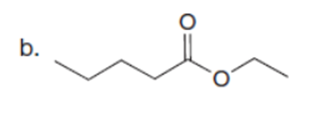
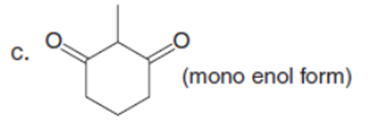
Q32.
Question: Both pentane-2,4-dione and ethyl acetoacetate have two carbonyl groups separated by a single carbon atom. Although an equilibrium mixture of pentane-2,4-dione tautomers contains 76% of the enol forms, an equilibrium mixture of ethyl acetoacetate tautomers contains only 8% of the enol forms. Suggest a reason for this difference.
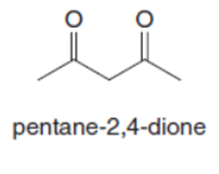
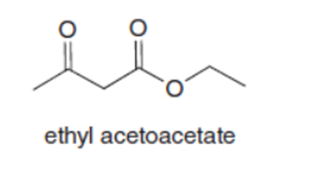
Q33.
Question: Which carbonyl compound in each pair exhibits the higher percentage of the enol tautomer?
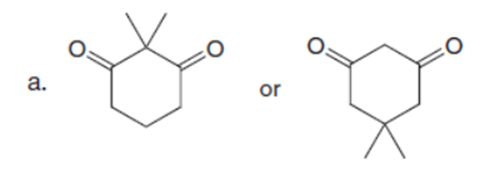
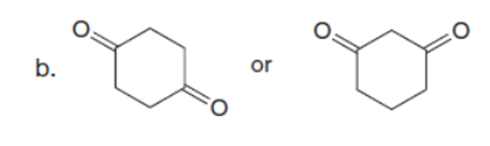
Q34.
Question: What hydrogen atoms in each compound have a pka≤ 25?
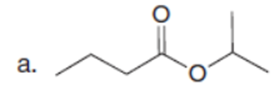
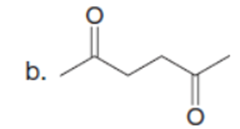
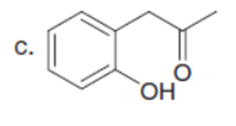
Q35.
Question: Rank the labeled protons in each compound in order of increasing acidity.
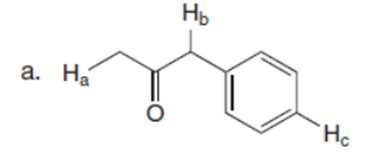
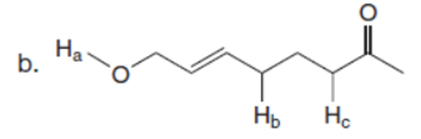
Q36.
Question:What is the major enolate (or carbanion) formed when each compound is treated with LDA?