Chapter 15: Radical Reactions
Q28.
Question: a) Draw all constitutional isomers formed by monochlorination of each alkane with Cl2and hν. (b) Draw the major monobromination product formed by heating each alkane with Br2.
Q29.
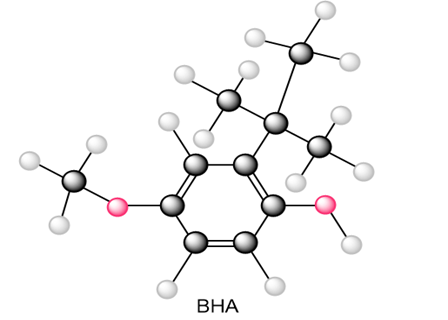
Question: Draw all resonance structures of the radical that results from abstraction of a hydrogen atom from the antioxidant BHA (butylated hydroxy anisole).
Q3.
Question: Draw the products formed when a chlorine atom reacts with each species.
a.
b.
c.
d. O2
Q30.
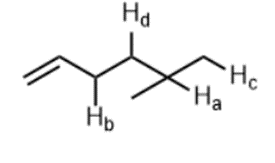
Question: With reference to the indicated C-H bonds in 2-methylbutane.
a. Rank the C-H bonds in order of increasing bond length.
b. Draw the radical resulting from cleavage of each C-H bond and classify it as 10,20, or 30
c.Rank the order of radicals in order of increasing stability.
Rank the C-H bonds in order of increasing ease of H abstractions in a radical halogenation reaction.
Q31.
Question: Rank the following radicals in order of increasing stability.
Q32.
Questions: Why is a benzylic C-H bond unusually weak?
Q33.
Questions: Rank the indicated hydrogen atoms in order of increasing ease of abstraction in a radical halogenation reaction.
Q34.
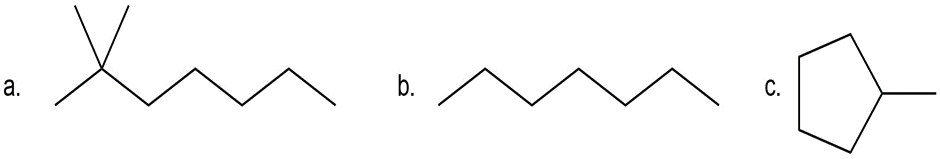
Question: Draw all constitutional isomers formed by monochlorination of each alkane with Cl2 and h.
Q35.
Question: What is the major monobromination product formed by heating each alkane with Br2 ?
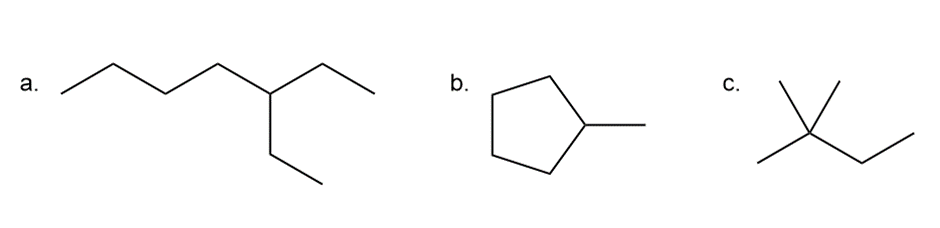
Q36.
Question: Five isomeric alkanes (A–E) having the molecular formula C6H14 are each treated with Cl2 + hv to give alkyl halides having molecular formula C6H13Cl . A yields five constitutional isomers. B yields four constitutional isomers. C yields two constitutional isomers. D yields three constitutional isomers, two of which possess stereo-genic centres. E yields three constitutional isomers, only one of which possesses a stereo-genic centre. Identify the structures of A–E.






