Chapter 15: Q32. (page 570)
Questions: Why is a benzylic C-H bond unusually weak?
Short Answer
Answer
Benzylic C-H bond is unusually weak because the cleavage of the C-H bond gives a 1o radical, which is stabilized by resonance.
Learning Materials
Features
Discover
Chapter 15: Q32. (page 570)
Questions: Why is a benzylic C-H bond unusually weak?
Answer
Benzylic C-H bond is unusually weak because the cleavage of the C-H bond gives a 1o radical, which is stabilized by resonance.
All the tools & learning materials you need for study success - in one app.
Get started for free
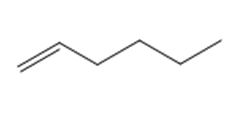
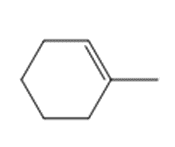
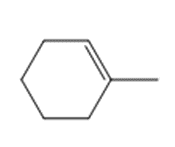
Question: Draw the product(s) formed when each alkene is treated with either [1] HBr alone; or [2] HBr in the presence of peroxides.
a.
b.
c.
Question: Calculate H0 for the rate-determining step of the reaction of CH4 with I2. Explain why this result illustrates that this reaction is extremely slow.
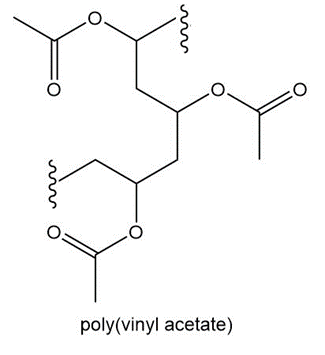
Question: (a)Draw the structure of polystyrene, the chapter-opening molecule, which is formed by polymerizing the monomer styrene, C6H5CH=CH2. (b) What monomer is used to form poly(vinyl acetate), a polymer used in paints and adhesives?
Question: Consider the following Bromination:
(CH)3 CH +Br2(CH3)3 CBr +HBr
(a) Calculate ΔH° for this reaction by using the bond dissociation energies in Table 6.2. (b) Draw out a stepwise mechanism for the reaction, including the initiation, propagation, and termination steps. (c) Calculate ΔH° for each propagation step. (d) Draw an energy diagram for the propagation steps. (e) Draw the structure of the transition state of each propagation step.
Questions: Compounds A and B are isomers having molecular formula C5H12. Heating A with Cl2 gives a single product of monohalogenation, whereas heating B under the same conditions forms three constitutional isomers. What are the structures of A and B?
What do you think about this solution?
We value your feedback to improve our textbook solutions.