Chapter 15: Q31. (page 570)
Question: Rank the following radicals in order of increasing stability.
Short Answer
Answer
The rank of the given radicals in the increasing order of stability is as follows:
Learning Materials
Features
Discover
Chapter 15: Q31. (page 570)
Question: Rank the following radicals in order of increasing stability.
Answer
The rank of the given radicals in the increasing order of stability is as follows:
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: When 3,3-dimethylbut-1-ene is treated with HBr alone, the major product is 2-bromo-2,3- dimethylbutane. When the same alkene is treated with HBr and peroxide, the sole product is 1-bromo-3,3-dimethylbutane. Explain these results by referring to the mechanisms.
Question: Draw the steps of the mechanism that converts vinyl chloride (CH2CHCl) into poly(vinyl chloride).
Question: What reagents are needed to convert 1-ethylcyclohexene into (a) 1-bromo-2-ethylcyclohexane;
(b) 1-bromo-1-ethylcyclohexane; (c) 1,2-dibromo-1-ethylcyclohexane?
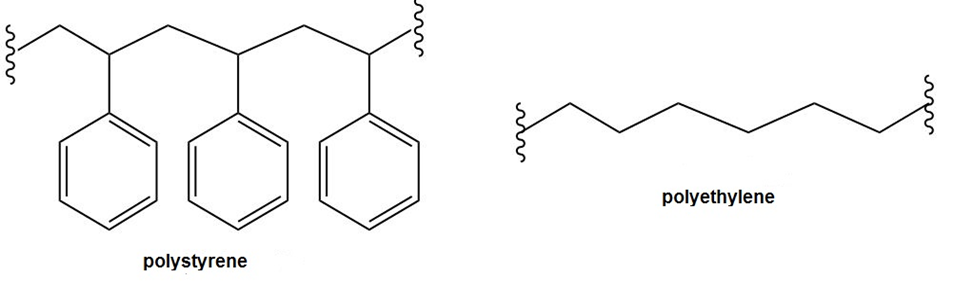
Question: Explain why polystyrene is much more readily oxidized by O2 in the air than polyethylene is. Which H’s in polystyrene are most easily abstracted and why?
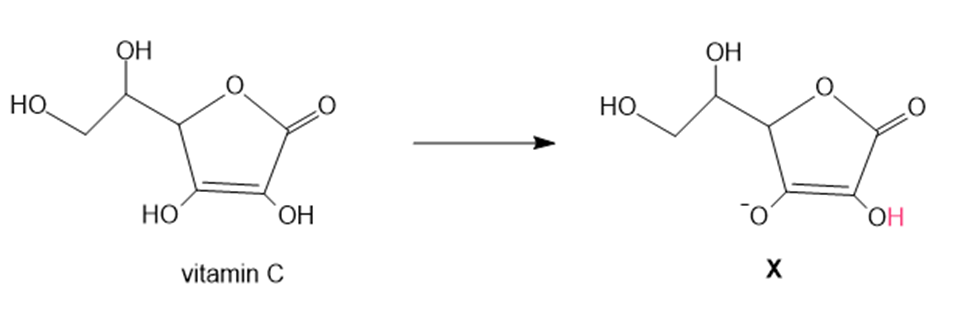
Question: In cells, vitamin C exists largely as its conjugate base X. X is an antioxidant because radicals formed in oxidation processes abstract the labeled H atom, forming a new radical that haltsoxidation. Draw the structure of the radical formed by H abstraction, and explain why this Hatom is most easily removed.
What do you think about this solution?
We value your feedback to improve our textbook solutions.