Chapter 15: Q27. (page 570)
Question: Draw the steps of the mechanism that converts vinyl chloride (CH2CHCl) into poly(vinyl chloride).
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 15: Q27. (page 570)
Question: Draw the steps of the mechanism that converts vinyl chloride (CH2CHCl) into poly(vinyl chloride).
Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
Questions: Why is a benzylic C-H bond unusually weak?
Question: Draw the products formed when each alkene is treated with NBS + hv .
a.
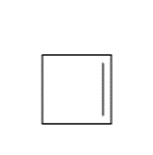
b.
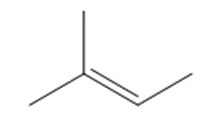
c.
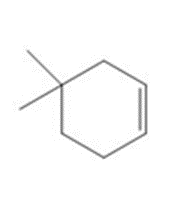
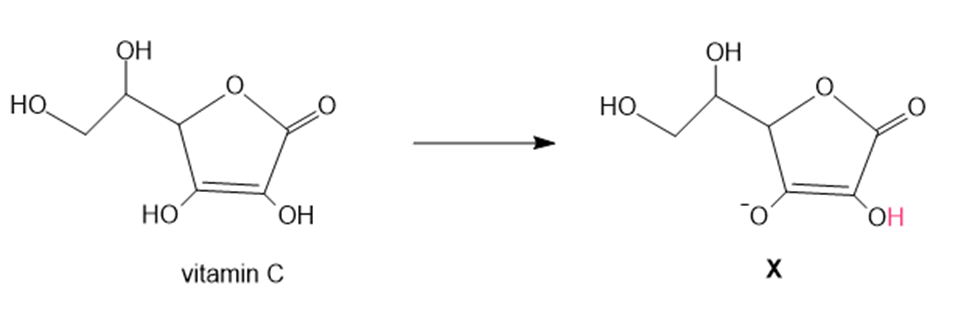
Question: In cells, vitamin C exists largely as its conjugate base X. X is an antioxidant because radicals formed in oxidation processes abstract the labeled H atom, forming a new radical that haltsoxidation. Draw the structure of the radical formed by H abstraction, and explain why this Hatom is most easily removed.
Question: Draw the product(s) formed when each alkene is treated with either [1] HBr alone; or [2] HBr in the presence of peroxides.
a.
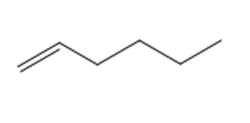
b.
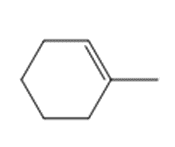
c.
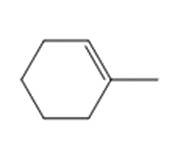
Questions: Compounds A and B are isomers having molecular formula C5H12. Heating A with Cl2 gives a single product of monohalogenation, whereas heating B under the same conditions forms three constitutional isomers. What are the structures of A and B?
What do you think about this solution?
We value your feedback to improve our textbook solutions.