Chapter 15: Q28. (page 570)
Question: a) Draw all constitutional isomers formed by monochlorination of each alkane with Cl2and hν. (b) Draw the major monobromination product formed by heating each alkane with Br2.
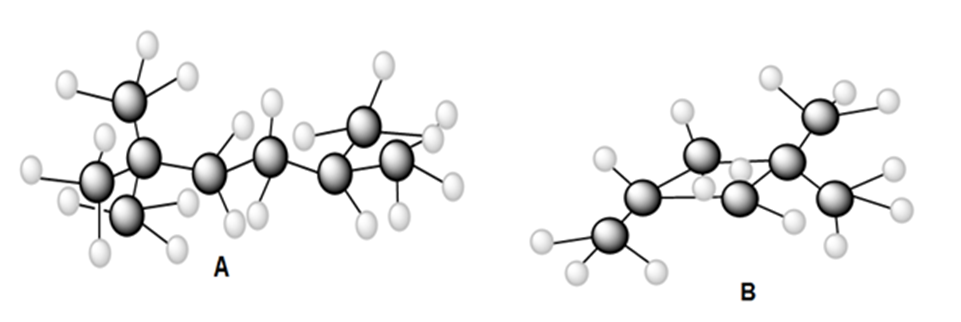
Short Answer
Answer
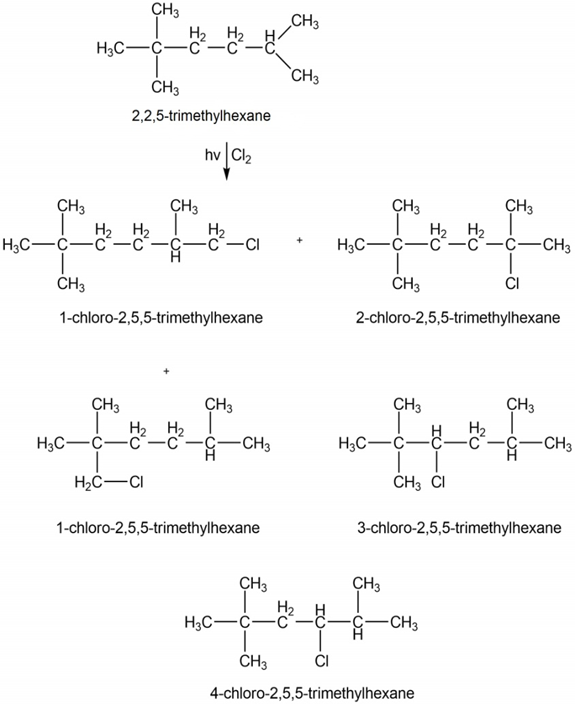
(a)
(b)
Learning Materials
Features
Discover
Chapter 15: Q28. (page 570)
Question: a) Draw all constitutional isomers formed by monochlorination of each alkane with Cl2and hν. (b) Draw the major monobromination product formed by heating each alkane with Br2.

Answer

(a)
(b)
All the tools & learning materials you need for study success - in one app.
Get started for free
Question:What reagents are needed to convert cyclopentene into
(a)bromocyclopentane;
(b) trans-1,2-dibromocyclopentane;
(c) 3-bromocyclopentene?
Consider the following Bromination:
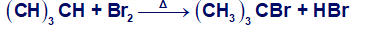
(e) Draw the structure of the transition state of each propagation step
Questions: Draw all constitutional isomers formed by monochlorination of each alkane.
a.
b.
c.
Question: Draw a stepwise mechanism for the following polymerization reaction
Question: (a)Hard contact lenses, which first became popular in the 1960s, were made by polymerizing methyl methacrylate [CH2=C(CH3)CO2CH3] to form poly(methyl methacrylate) (PMMA). Draw the structure of PMMA. (b) More comfortable softer contact lenses introduced in the 1970swere made by polymerizing hydroxyethyl methacrylate [CH2=C(CH3)CO2CH2CH2OH] to form
poly(hydroxyethyl methacrylate) (poly-HEMA). Draw the structure of poly-HEMA. Since neither polymer allows oxygen from the air to pass through to the retina, newer contact lenses that are both comfortable and oxygen-permeable have now been developed.
What do you think about this solution?
We value your feedback to improve our textbook solutions.