Chapter 15: Q4. (page 570)
Questions: Draw all constitutional isomers formed by monochlorination of each alkane.
a.
b.
c.
Short Answer
Answer
a.
b.
c.
Learning Materials
Features
Discover
Chapter 15: Q4. (page 570)
Questions: Draw all constitutional isomers formed by monochlorination of each alkane.
a.
b.
c.
Answer
a.
b.
c.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: The triphenylmethyl radical is an unusual persistent radical present in solution in equilibrium with its dimer. For 70 years the dimer was thought to be hexaphenylethane, but in 1970, NMR data showed it to be A.
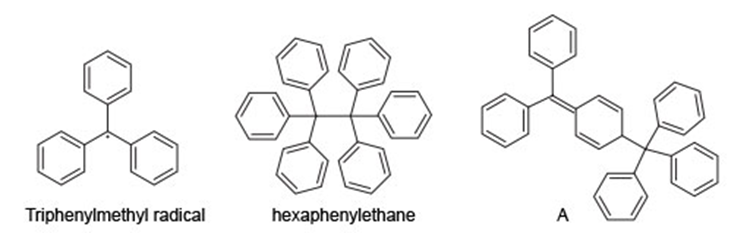
a. Why is the triphenylmethyl radical more stable than most other radicals?
b. Use curved arrow notation to show how two triphenylmethyl radicals dimerize to form A.
c. Propose a reason for the formation of A rather than hexaphenylethane.d. How could 1 H and 13C NMR spectroscopy be used to distinguish between hexaphenylethane and A?
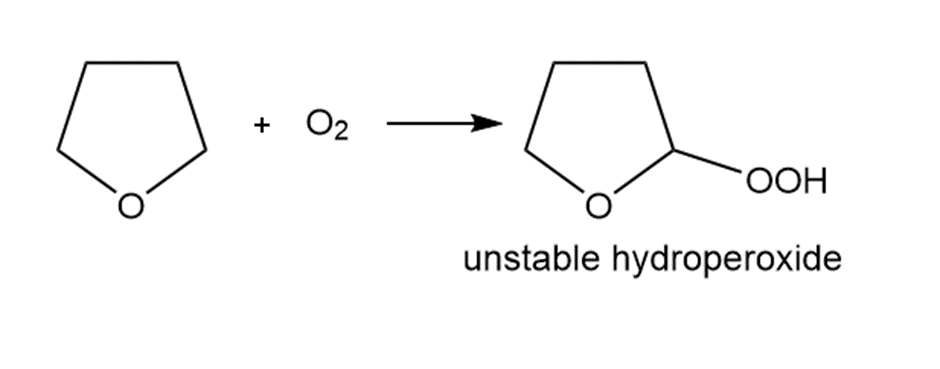
Question. Ethers are oxidized with to form hydroperoxides that decompose violently when heated. Draw a stepwise mechanism for this reaction.
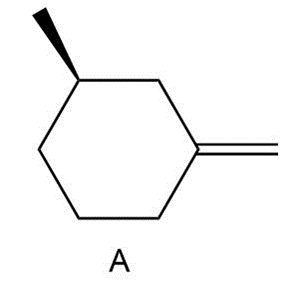
Question: Draw the six products (including stereoisomers) formed when A is treated with NBS + hv.
Question: What reagents are needed to convert 1-ethylcyclohexene into (a) 1-bromo-2-ethylcyclohexane;
(b) 1-bromo-1-ethylcyclohexane; (c) 1,2-dibromo-1-ethylcyclohexane?
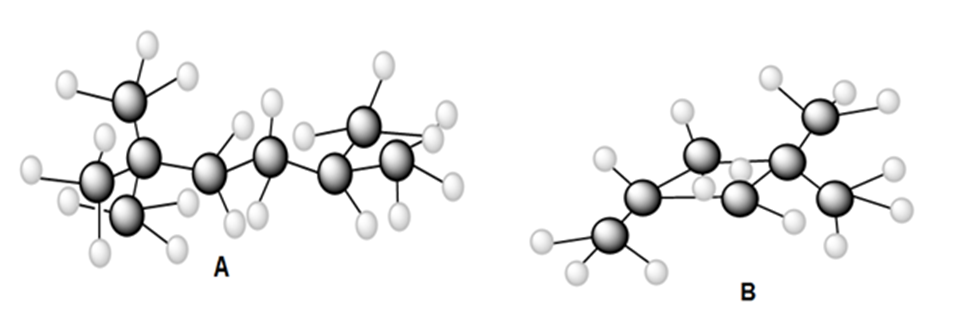
Question: a) Draw all constitutional isomers formed by monochlorination of each alkane with Cl2and hν. (b) Draw the major monobromination product formed by heating each alkane with Br2.
What do you think about this solution?
We value your feedback to improve our textbook solutions.