Chapter 15: Q64. (page 570)
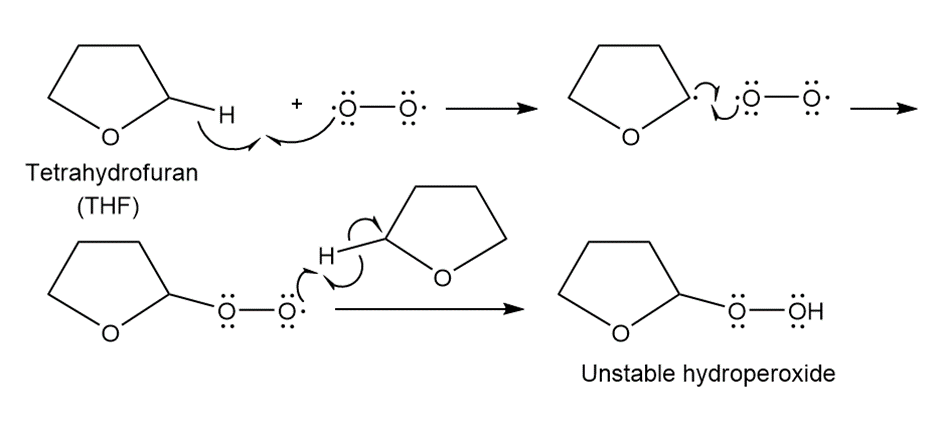
Question. Ethers are oxidized with to form hydroperoxides that decompose violently when heated. Draw a stepwise mechanism for this reaction.
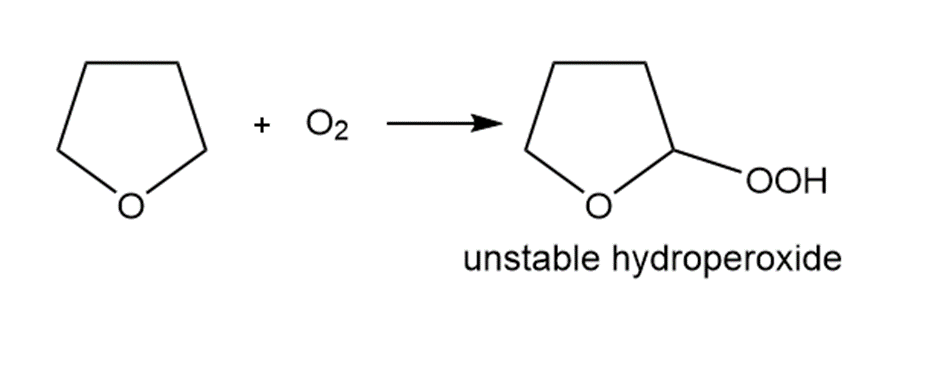
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 15: Q64. (page 570)
Question. Ethers are oxidized with to form hydroperoxides that decompose violently when heated. Draw a stepwise mechanism for this reaction.

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Consider the following Bromination:
(CH)3 CH +Br2(CH3)3 CBr +HBr
(a) Calculate ΔH° for this reaction by using the bond dissociation energies in Table 6.2. (b) Draw out a stepwise mechanism for the reaction, including the initiation, propagation, and termination steps. (c) Calculate ΔH° for each propagation step. (d) Draw an energy diagram for the propagation steps. (e) Draw the structure of the transition state of each propagation step.
Question: Draw the products of each reaction.
a.
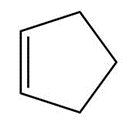
b.
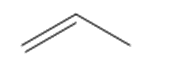
c.
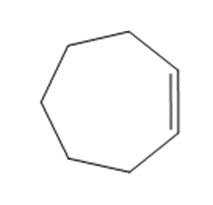
Question: Draw the major product formed when each cycloalkane is heated with Br2.
a.
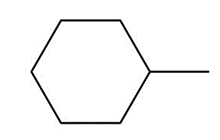
b.
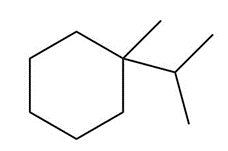
c.
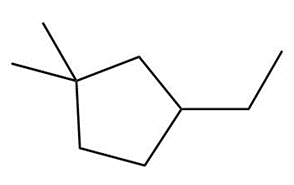
d.
Question: In the presence of a radical initiator (Z* ), tributyltin hydride ( R3SnH,R=CH3CH2CH2CH2 ) reduces alkyl halides to alkanes: R'X+R3SnHR'H+R3SnX. The mechanism consists of a radical chain process with an intermediate tin radical:
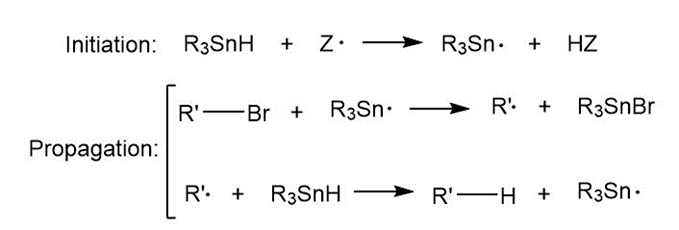
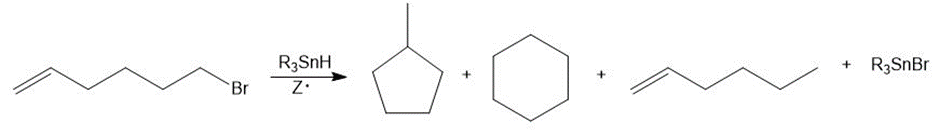
This reaction has been employed in many radical cyclization reactions. Draw a stepwise mechanism for the following reaction.
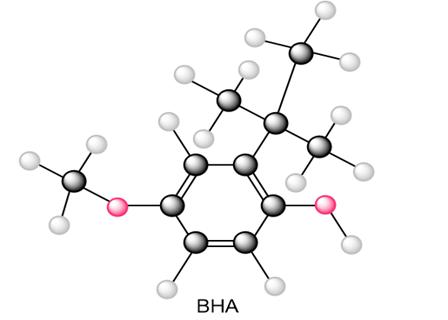
Question: Draw all resonance structures of the radical that results from abstraction of a hydrogen atom from the antioxidant BHA (butylated hydroxy anisole).
What do you think about this solution?
We value your feedback to improve our textbook solutions.