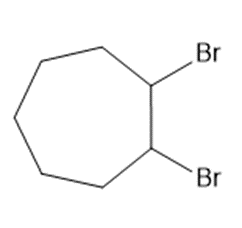
Chapter 15: Q17. (page 570)
Question: Draw the products of each reaction.
a.
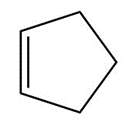
b.
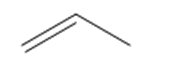
c.

Short Answer
Answer
a.
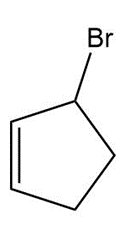
b.
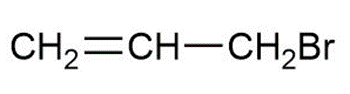
c.
Learning Materials
Features
Discover
Chapter 15: Q17. (page 570)
Question: Draw the products of each reaction.
a.

b.

c.

Answer
a.

b.

c.

All the tools & learning materials you need for study success - in one app.
Get started for free
Consider the following Bromination:
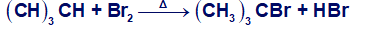
(e) Draw the structure of the transition state of each propagation step
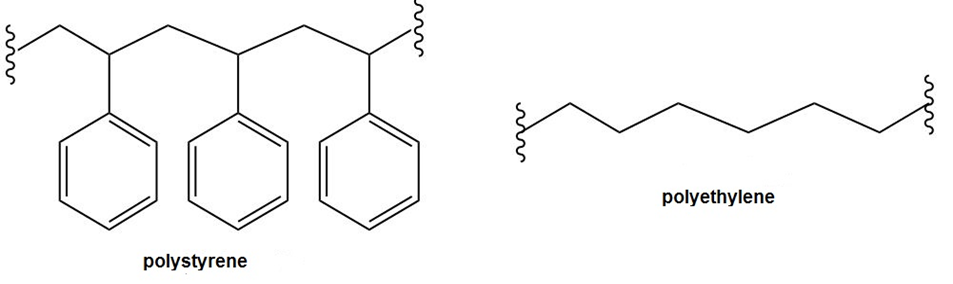
Question: Explain why polystyrene is much more readily oxidized by O2 in the air than polyethylene is. Which H’s in polystyrene are most easily abstracted and why?
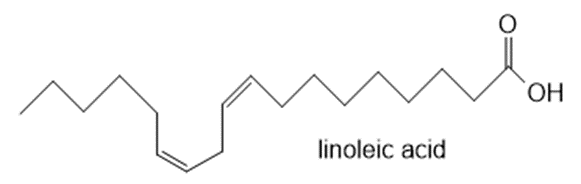
Question: Which C-H bond is most readily cleaved in linoleic acid? Draw all possible resonance structures for the resulting radical. Draw all the hydroperoxides formed by the reaction of this resonance stabilized radical with O2.
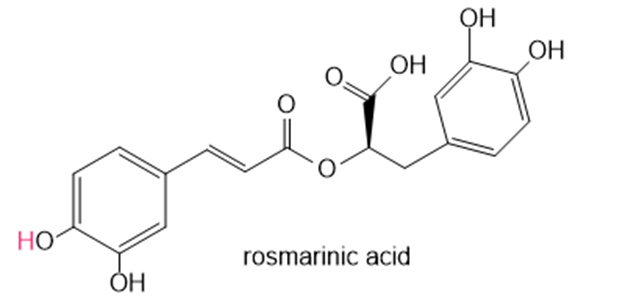
Question: Rosmarinic acid is an antioxidant isolated from rosemary. Draw resonance structures for the radical that results from removal of the labeled H atom in rosmarinic acid.
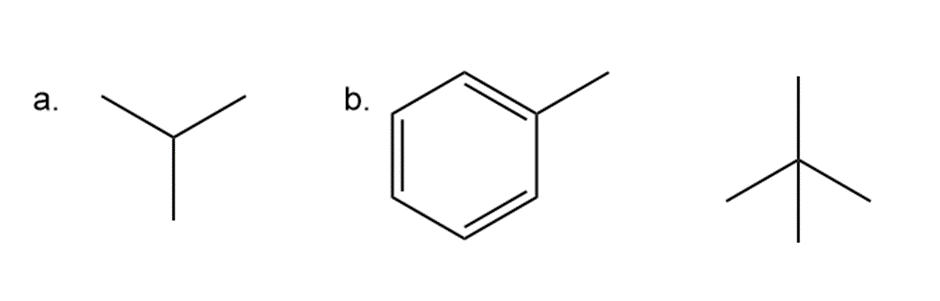
Question: Draw the products of radical chlorination and bromination of each compound. For which compounds is a single constitutional isomer formed for both reactions? What must be true about the structure of a reactant for both reactions to form a single product?
What do you think about this solution?
We value your feedback to improve our textbook solutions.