Chapter 15: Q18. (page 570)
Question: Draw all constitutional isomers formed when each alkene is treated with NBS + hν.
a.
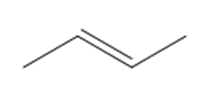
b.
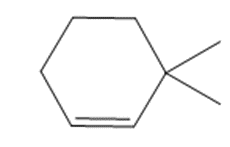
c.
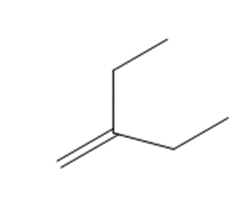
Short Answer
Answer
a.
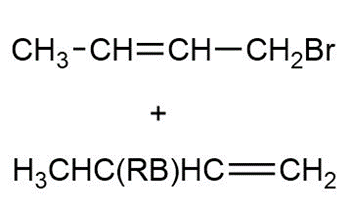
b.
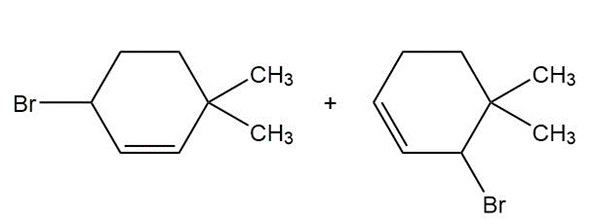
c.
Learning Materials
Features
Discover
Chapter 15: Q18. (page 570)
Question: Draw all constitutional isomers formed when each alkene is treated with NBS + hν.
a.

b.

c.

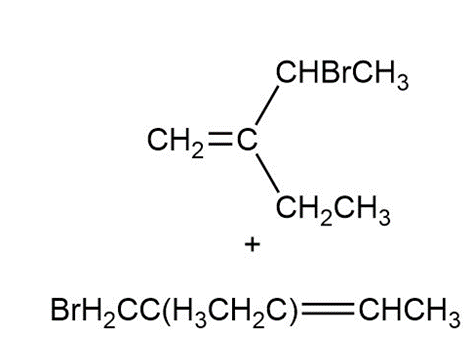
Answer
a.
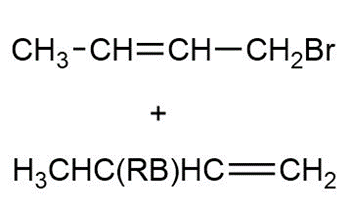
b.

c.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Draw all resonance structures of the radical that results from abstraction of a hydrogen atom from the antioxidant BHA (butylated hydroxy anisole).
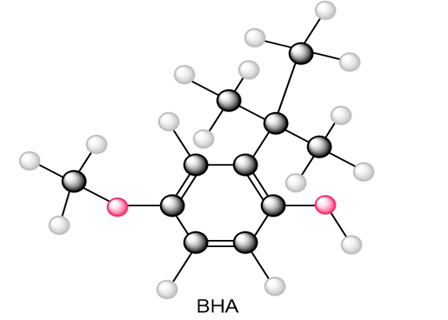
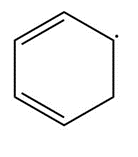
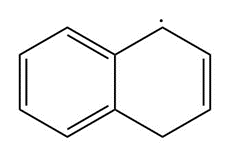
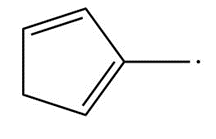
Question: Draw resonance structures for each radical.
a.
b.
c.
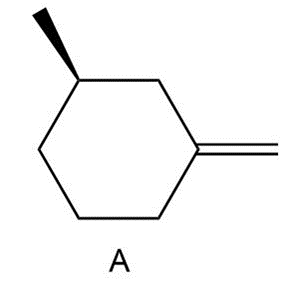
Question: Draw the six products (including stereoisomers) formed when A is treated with NBS + hv.
Question: When 3,3-dimethylbut-1-ene is treated with HBr alone, the major product is 2-bromo-2,3- dimethylbutane. When the same alkene is treated with HBr and peroxide, the sole product is 1-bromo-3,3-dimethylbutane. Explain these results by referring to the mechanisms.
Question: With reference to the indicated C-H bonds in 2-methylbutane.
a. Rank the C-H bonds in order of increasing bond length.
b. Draw the radical resulting from cleavage of each C-H bond and classify it as 10,20, or 30
c.Rank the order of radicals in order of increasing stability.
Rank the C-H bonds in order of increasing ease of H abstractions in a radical halogenation reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.