Chapter 15: Q42. (page 570)
Question: Draw resonance structures for each radical.
a.
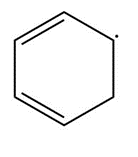
b.
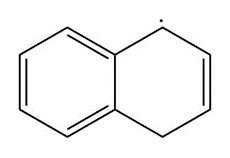
c.
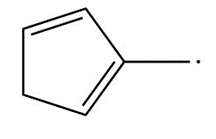
Short Answer
Answer
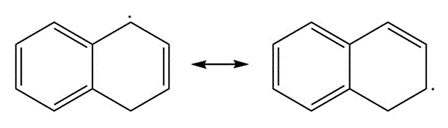
a.
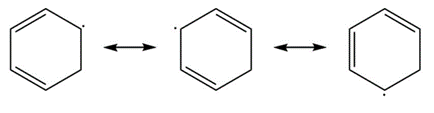
Resonance in a
b.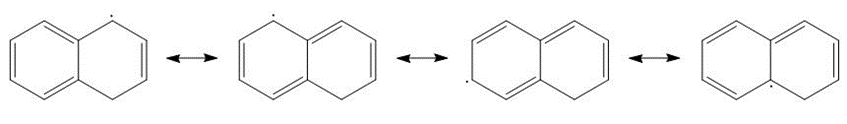
Resonance possibility 1 in b
Resonance possibility 2 in b
c.
Resonance in c
Learning Materials
Features
Discover
Chapter 15: Q42. (page 570)
Question: Draw resonance structures for each radical.
a.

b.

c.

Answer
a.

Resonance in a
b.
Resonance possibility 1 in b

Resonance possibility 2 in b
c.
Resonance in c
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: With reference to the indicated C-H bonds in 2-methylbutane.
a. Rank the C-H bonds in order of increasing bond length.
b. Draw the radical resulting from cleavage of each C-H bond and classify it as 10,20, or 30
c.Rank the order of radicals in order of increasing stability.
Rank the C-H bonds in order of increasing ease of H abstractions in a radical halogenation reaction.
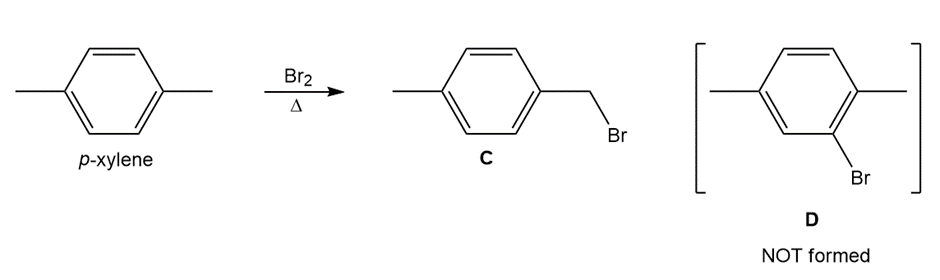
Question: Explain why radical bromination of p-xylene forms C rather than D.
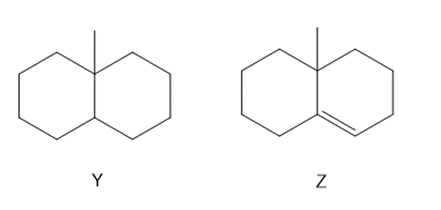
Question: a. What product(s) (excluding stereoisomers) are formed when Y is heated with Cl2 ?
b. What product(s) (excluding stereoisomers) are formed when Y is heated with Br2?
c. What steps are needed to convert Y to the alkene Z?
Question: Devise a synthesis of each compound using CH3CH3 as the only source of carbon atoms. You may use any other required organic or inorganic reagents.
a.
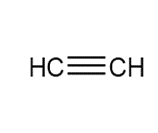
b.
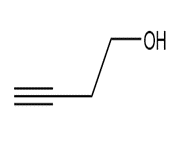
c.
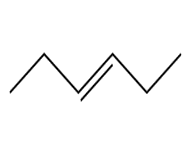
d.
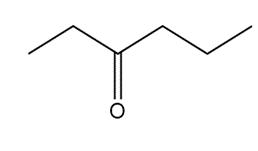
Question: What products are formed from monochlorination of (R)-2-bromobutane at C1 and C4? Assign R and S designations to each stereogenic center.
What do you think about this solution?
We value your feedback to improve our textbook solutions.