Chapter 15: Q22. (page 570)
Question: Rosmarinic acid is an antioxidant isolated from rosemary. Draw resonance structures for the radical that results from removal of the labeled H atom in rosmarinic acid.

Short Answer
Answer
Learning Materials
Features
Discover
Chapter 15: Q22. (page 570)
Question: Rosmarinic acid is an antioxidant isolated from rosemary. Draw resonance structures for the radical that results from removal of the labeled H atom in rosmarinic acid.

Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
Question:What reagents are needed to convert cyclopentene into
(a)bromocyclopentane;
(b) trans-1,2-dibromocyclopentane;
(c) 3-bromocyclopentene?
Question: Calculate H0 for the rate-determining step of the reaction of CH4 with I2. Explain why this result illustrates that this reaction is extremely slow.
Question: Classify each radical as Primary , secondary or tertiary carbon radicals .
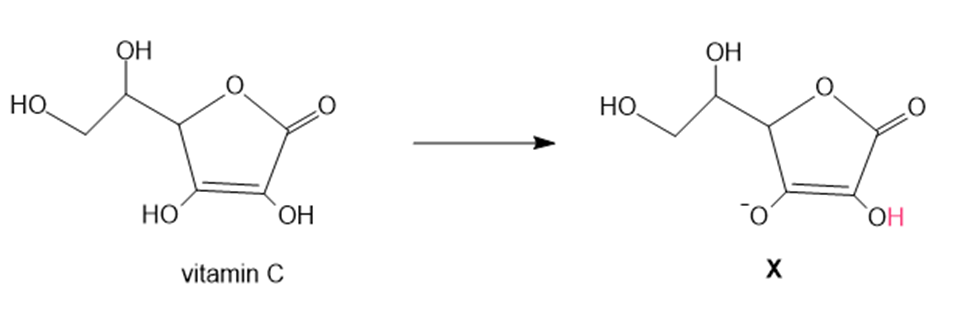
Question: In cells, vitamin C exists largely as its conjugate base X. X is an antioxidant because radicals formed in oxidation processes abstract the labeled H atom, forming a new radical that haltsoxidation. Draw the structure of the radical formed by H abstraction, and explain why this Hatom is most easily removed.
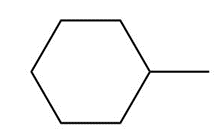
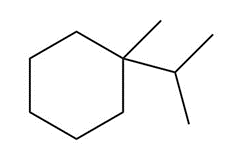
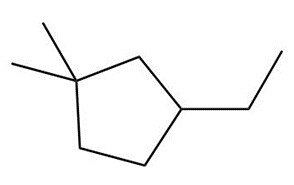
Question: Draw the major product formed when each cycloalkane is heated with Br2.
a.
b.
c.
d.
What do you think about this solution?
We value your feedback to improve our textbook solutions.