Chapter 15: Q1. (page 570)
Question: Classify each radical as Primary , secondary or tertiary carbon radicals .
Short Answer
Answer
a. carbon radical
b. carbon radical
c. carbon radical
d. localid="1648713761224" carbon radical
Learning Materials
Features
Discover
Chapter 15: Q1. (page 570)
Question: Classify each radical as Primary , secondary or tertiary carbon radicals .
Answer
a. carbon radical
b. carbon radical
c. carbon radical
d. localid="1648713761224" carbon radical
All the tools & learning materials you need for study success - in one app.
Get started for free
Questions: Compounds A and B are isomers having molecular formula C5H12. Heating A with Cl2 gives a single product of monohalogenation, whereas heating B under the same conditions forms three constitutional isomers. What are the structures of A and B?
Question: Draw all constitutional isomers formed by monochlorination of each alkane with Cl2 and h.
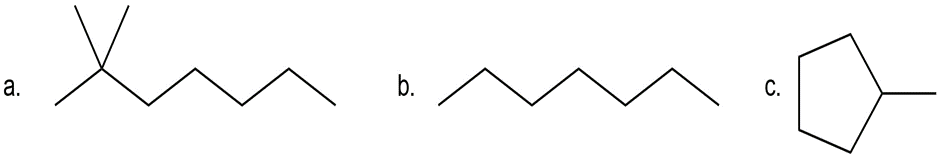
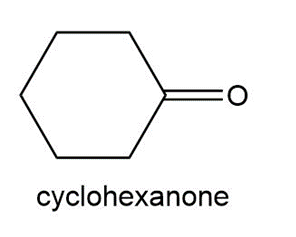
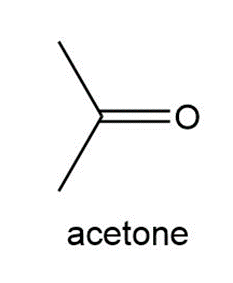
Question:Treatment of a hydrocarbon A (molecular formula C9H18) with Br2 in the presence of light forms alkyl halides B and C, both having molecular formula C9H17Br. Reaction of either B or C with KOC (CH3)3 forms compound D ( C9H16) as the major product. Ozonolysis of D forms cyclohexanone and acetone. Identify the structures of A–D.
Question: (a) Draw the products (including stereoisomers) formed when 2-methylhex-2-ene is treated with HBr in the presence of peroxides. (b) Draw the products (including stereoisomers) formed when (S)-2,4-dimethylhex-2-ene is treated with HBr and peroxides under similar conditions.
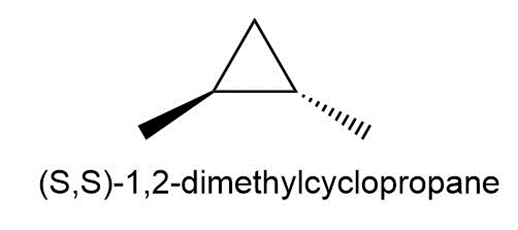
Question:Draw all the monochlorination products that are formed from (S,S)-1,2-dimethylcyclopropane.
What do you think about this solution?
We value your feedback to improve our textbook solutions.