Chapter 15: Q50. (page 570)
Question:Draw all the monochlorination products that are formed from (S,S)-1,2-dimethylcyclopropane.
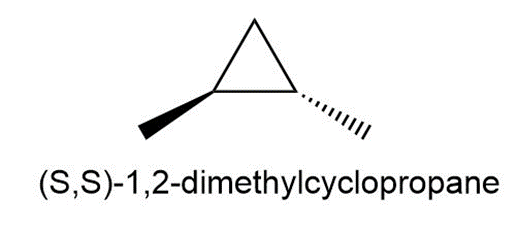
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 15: Q50. (page 570)
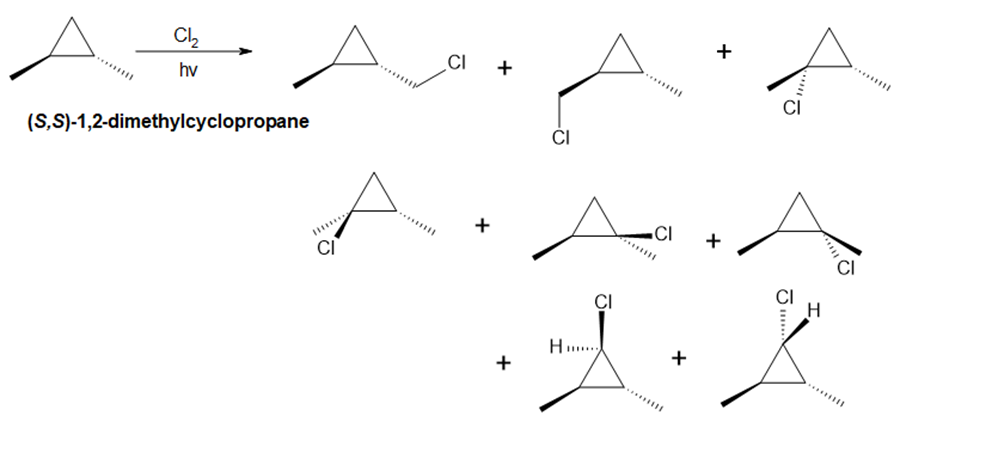
Question:Draw all the monochlorination products that are formed from (S,S)-1,2-dimethylcyclopropane.

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
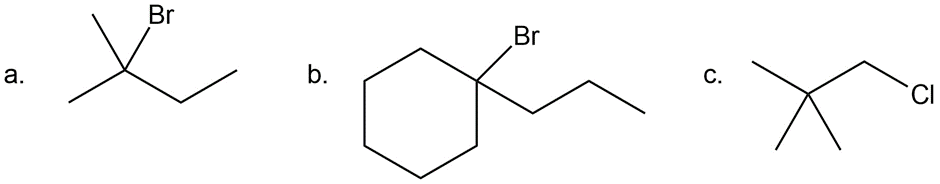
Question: What alkane is needed to make each alkyl halide by radical halogenation?
Question: Reaction of (CH3 )3 CH with Cl2 forms two products: (CH3 )2 CHCH2Cl (63%) and (CH3)3 CCl (37%). Why is the major product formed by cleavage of the stronger 1° C-H bond?
Question: Draw the major product formed when each cycloalkane is heated with Br2.
a.
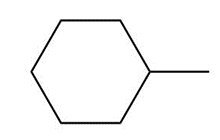
b.
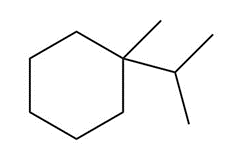
c.
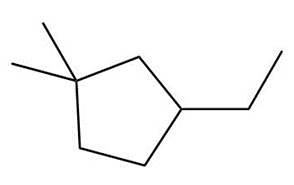
d.
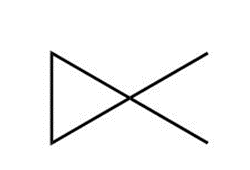
Question: Draw the products formed when a chlorine atom reacts with each species.
a.
b.
c.
d. O2
Question: When 3,3-dimethylbut-1-ene is treated with HBr alone, the major product is 2-bromo-2,3- dimethylbutane. When the same alkene is treated with HBr and peroxide, the sole product is 1-bromo-3,3-dimethylbutane. Explain these results by referring to the mechanisms.
What do you think about this solution?
We value your feedback to improve our textbook solutions.