Chapter 15: Q77. (page 570)
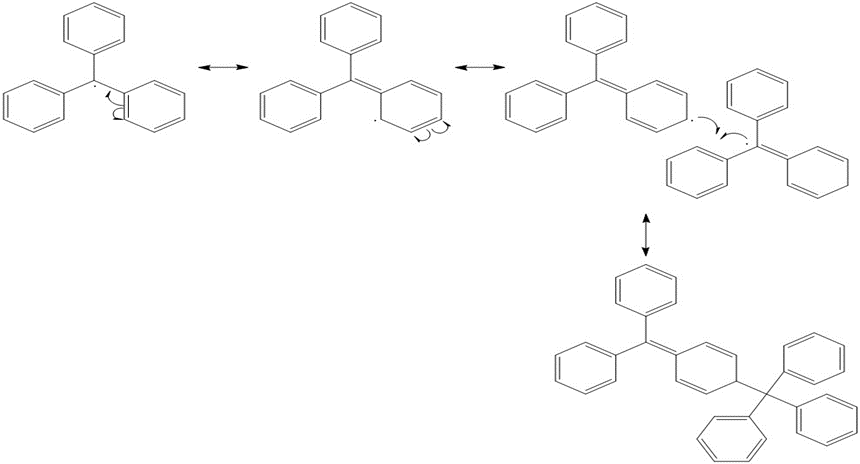
Question: The triphenylmethyl radical is an unusual persistent radical present in solution in equilibrium with its dimer. For 70 years the dimer was thought to be hexaphenylethane, but in 1970, NMR data showed it to be A.
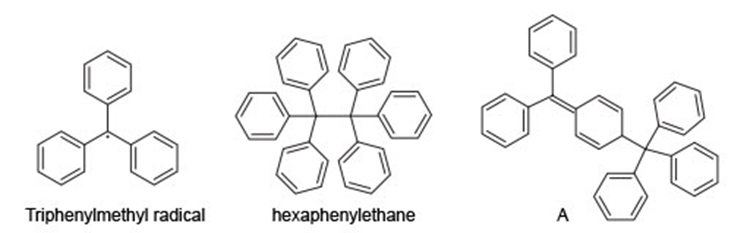
a. Why is the triphenylmethyl radical more stable than most other radicals?
b. Use curved arrow notation to show how two triphenylmethyl radicals dimerize to form A.
c. Propose a reason for the formation of A rather than hexaphenylethane.d. How could 1 H and 13C NMR spectroscopy be used to distinguish between hexaphenylethane and A?
Short Answer
Answer
a. Triphenylmethyl radical has more number of resonance structures.
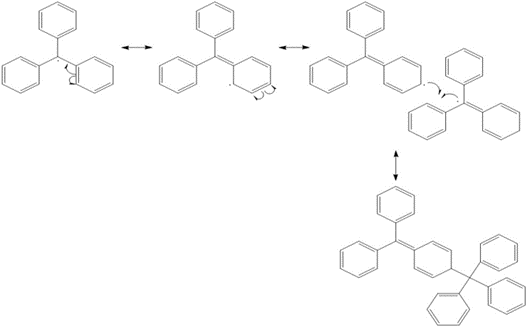
b. Dimerization of triphenyl methyl radical
c. Hexaphenyl ethane has more steric hindrance than Compound A.
d. Due to the quinonoid ring in compound A, the proton and carbon-13 NMR of compound A will have more peaks.