Chapter 10: Q.27. (page 414)
Question: Draw the products formed when each alkene is treated with followed by . Include the stereochemistry at all stereogenic centers.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 10: Q.27. (page 414)
Question: Draw the products formed when each alkene is treated with followed by . Include the stereochemistry at all stereogenic centers.
Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Iejimalide B, an anticancer agent with a 24-membered ring, is isolated from a tunicate found off Ie Island in Okinawa.
(a) Label each double bond in iejimalide B as Eor Z.
(b) Label each tetrahedral stereogenic center as Ror S.
(c) How many stereoisomers are possible for iejimalide B?
Question: Devise a synthesis of each product from the given starting material. More than one step is required.
a.
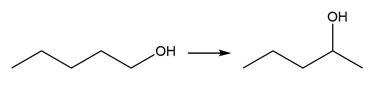
b.
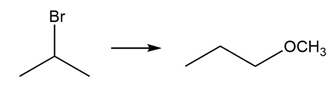
c.
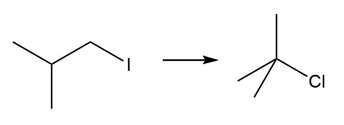
d.
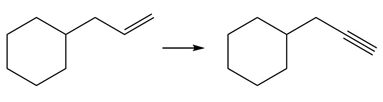
e.
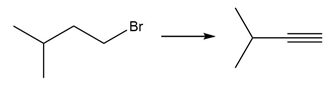
f.
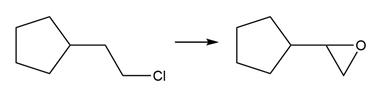
Question: What three alkenes (excluding stereoisomers) can be used to prepare 3-chloro-3-methylhexane by addition of HCl?
Question: Draw the six alkenes of the molecular formula C5H10. Label one pair of diastereomers.
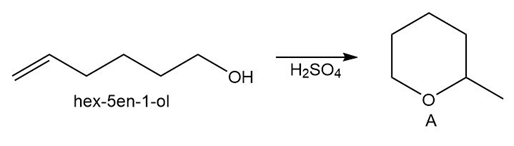
Question: Draw a stepwise mechanism for the conversion of hex-5-en-1-ol to the cyclic ether A.
What do you think about this solution?
We value your feedback to improve our textbook solutions.