Chapter 10: Q.28. (page 414)
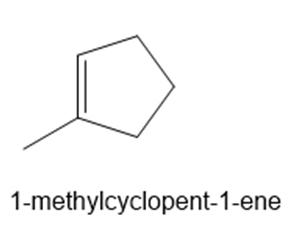
Question: What alkene can be used to prepare each alcohol as the exclusive product of a two-step hydroboration–oxidation sequence?
Short Answer
Answer
a.
Learning Materials
Features
Discover
Chapter 10: Q.28. (page 414)
Question: What alkene can be used to prepare each alcohol as the exclusive product of a two-step hydroboration–oxidation sequence?
Answer
a.

All the tools & learning materials you need for study success - in one app.
Get started for free
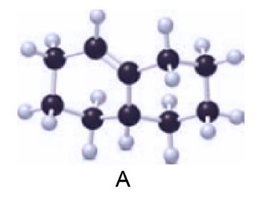
Question: (a) Label the carbon–carbon double bond in A as E or Z. (b) Draw the products (including stereoisomers) formed when A is treated with in the presence of .
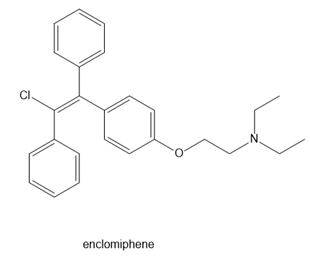
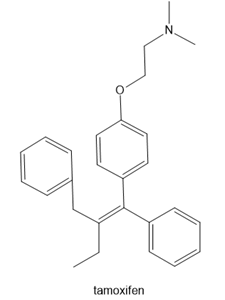
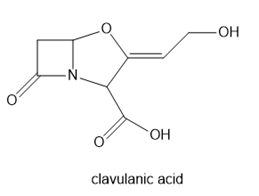
Question: Label the alkene in each drug as E or Z. Enclomiphene is one component of the fertility drug Clomid. Tamoxifen is an anticancer drug. Clavulanic acid is sold in combination with the antibiotic amoxicillin under the trade name Augmentin.
a
b.
c.
Question: (a) Draw the structure of (1E,4R)-1,4-dimethylcyclodecene. (b) Draw the enantiomer and name it, including its E,Z and R,S prefixes. (c) Draw two diastereomers and name them, including the E,Z and R,S prefixes.
Question: What alkylborane is formed from hydroboration of each alkene?
Question: Which alcohols can be prepared as a single product by hydroboration–oxidation of an alkene? Which alcohols can be prepared as a single product by the acid-catalyzed addition of H2O to an alkene?
a.
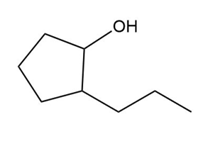
b.
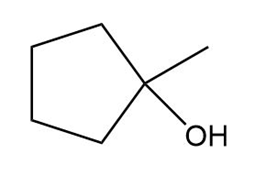
c.
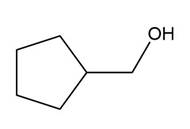
d.
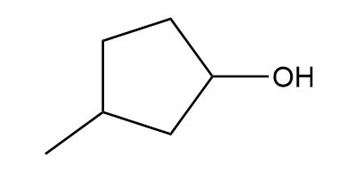
What do you think about this solution?
We value your feedback to improve our textbook solutions.