Chapter 10: PROBLEM 10.48 (page 422)
Question: Which alcohols can be prepared as a single product by hydroboration–oxidation of an alkene? Which alcohols can be prepared as a single product by the acid-catalyzed addition of H2O to an alkene?
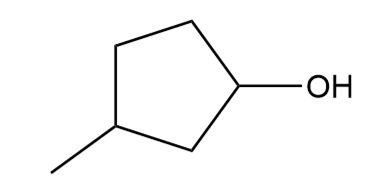
a.
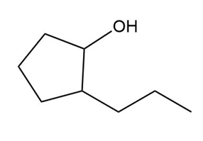
b.
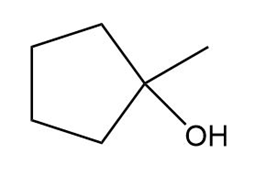
c.
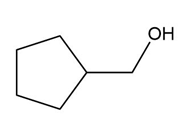
d.
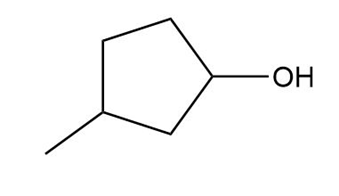
Short Answer
Answer
��.������Dz��ǰ����پ��Dz�–o�澱�岹�پ��Dz�.
b.Acid-catalyzed addition.
c.Acid-catalyzed addition.
d. ������Dz��ǰ����پ��Dz�–o�澱�岹�پ��Dz� and acid-catalyzed addition.
 ������Dz��ǰ����پ��Dz�–o�澱�岹�پ��Dz�
������Dz��ǰ����پ��Dz�–o�澱�岹�پ��Dz� Acid-catalyzed addition
Acid-catalyzed addition Acid-catalyzed addition
Acid-catalyzed addition