Chapter 10: PROBLEM 10.49 (page 422)
Question: Draw the constitutional isomer formed in each reaction.
a.
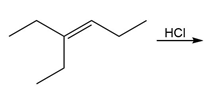
b.
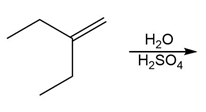
c.
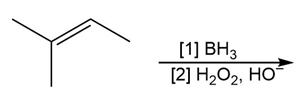
d.
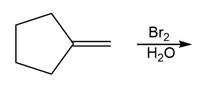
e.
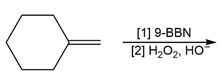
f.
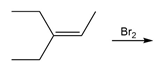
Short Answer
Answer
a.
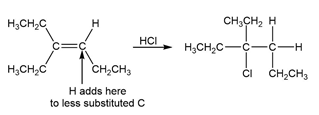
b.
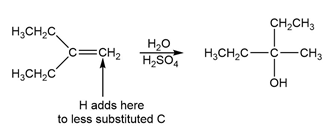
c.
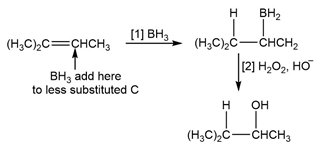
d.
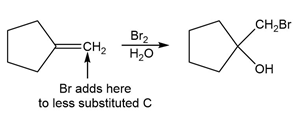
e.
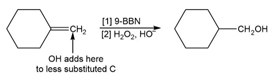
f.
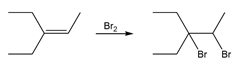
Learning Materials
Features
Discover
Chapter 10: PROBLEM 10.49 (page 422)
Question: Draw the constitutional isomer formed in each reaction.
a.

b.

c.

d.

e.
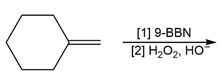
f.

Answer
a.

b.

c.

d.

e.

f.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: How many degrees of unsaturation are present in each compound?
Question: Draw the isomers for the following compounds and then name each one:
a. 2-methyl-2,4-hexadiene
b. 2,4-heptadiene
c. 1,3-pentadiene
Question: (a) Label the carbon–carbon double bond in A as E or Z. (b) Draw the products (including stereoisomers) formed when A is treated with in the presence of .
Question: Draw the constitutional isomer formed when the following alkenes are treated with each set of reagents: [1] ; or [2] followed by .
Question: (a) Draw the structure of (1E,4R)-1,4-dimethylcyclodecene. (b) Draw the enantiomer and name it, including its E,Z and R,S prefixes. (c) Draw two diastereomers and name them, including the E,Z and R,S prefixes.
What do you think about this solution?
We value your feedback to improve our textbook solutions.