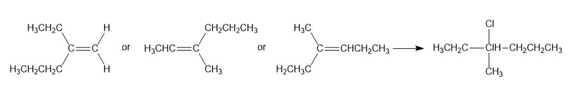
Chapter 10: PROBLEM 10.50 (page 422)
Question: What three alkenes (excluding stereoisomers) can be used to prepare 3-chloro-3-methylhexane by addition of HCl?
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 10: PROBLEM 10.50 (page 422)
Question: What three alkenes (excluding stereoisomers) can be used to prepare 3-chloro-3-methylhexane by addition of HCl?
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: (a) Draw the structure of (1E,4R)-1,4-dimethylcyclodecene. (b) Draw the enantiomer and name it, including its E,Z and R,S prefixes. (c) Draw two diastereomers and name them, including the E,Z and R,S prefixes.
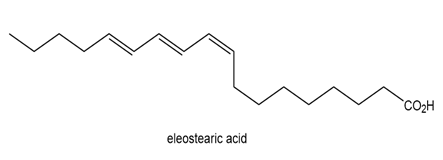
Question: Eleostearic acid is an unsaturated fatty acid obtained from the seeds of the tung oil tree (Aleurites fordii), a deciduous tree native to China.
(a) Draw the structure of a stereoisomer that has a higher melting point than eleostearic acid.
(b) Draw the structure of a stereoisomer that has a lower melting point.
Question: Draw the products formed when each alkene is treated with followed by . Include the stereochemistry at all stereogenic centers.
Question: Use the Hammond postulate to explain why reacts faster than in electrophilic addition of HX.
Question: When buta-1,3-diene (CH2=CH-CH=CH2) is treated with HBr, two constitutional isomers are formed CH3CHBr-CH=CH2and Br-CH2CH=CHCH2. Draw a stepwise mechanism that accounts for the formation of both products.
What do you think about this solution?
We value your feedback to improve our textbook solutions.