Chapter 10: Q.16. (page 401)
Question: Use the Hammond postulate to explain why reacts faster than in electrophilic addition of HX.
Short Answer
Answer
generates a carbocation and imparts greater stability. Hence, reacts faster.
Learning Materials
Features
Discover
Chapter 10: Q.16. (page 401)
Question: Use the Hammond postulate to explain why reacts faster than in electrophilic addition of HX.
Answer
generates a carbocation and imparts greater stability. Hence, reacts faster.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: How many degrees of unsaturation are present in each compound?
Question: Using cis- and trans-hex-3-ene, demonstrate that the addition of HCl is not a stereospecific reaction. Draw the structure of the stereoisomers formed from each alkene.
Question: Calculate the number of degrees of unsaturation for each molecular formula.
a. C6H8
b. C40H56
c. C10H16O2
d. C8H9Br
e. C8H9ClO
f. C7H11N
g. C4H8BrN
h. C10H10ClNO
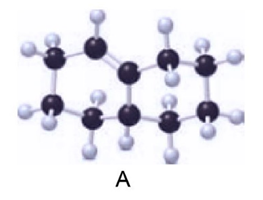
Question: (a) Label the carbon–carbon double bond in A as E or Z. (b) Draw the products (including stereoisomers) formed when A is treated with in the presence of .
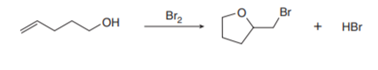
Question: Bromoetherification, the addition of the elements of Br and OR to a double bond, is a common method for constructing rings containing oxygen atoms. This reaction has been used in the synthesis of the polyether antibiotic monensin (Problem 21.37). Draw a stepwise mechanism for the following intramolecular bromoetherification reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.