Chapter 10: Q.15. (page 401)
Question: Draw the products formed when each alkene is treated with HCl.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 10: Q.15. (page 401)
Question: Draw the products formed when each alkene is treated with HCl.
Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Draw all stereoisomers formed in each reaction.
Question: Draw the products of each reaction using the two-part strategy from Sample Problem 10.7.
Question: Treatment of 3-methylcyclohexene with HCl yields two products, 1-chloro-3-methylcyclohexane and 1-chloro-1-methylcyclohexane. Draw a mechanism to explain this result.
Question: What alkene can be used to prepare each alcohol as the exclusive product of a two-step hydroboration–oxidation sequence?
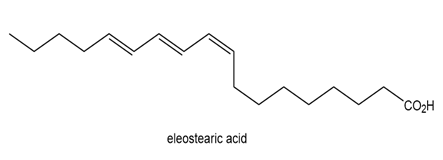
Question: Eleostearic acid is an unsaturated fatty acid obtained from the seeds of the tung oil tree (Aleurites fordii), a deciduous tree native to China.
(a) Draw the structure of a stereoisomer that has a higher melting point than eleostearic acid.
(b) Draw the structure of a stereoisomer that has a lower melting point.
What do you think about this solution?
We value your feedback to improve our textbook solutions.