Chapter 10: Q.29. (page 415)
Question: Draw the constitutional isomer formed when the following alkenes are treated with each set of reagents: [1] ; or [2] followed by .
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 10: Q.29. (page 415)
Question: Draw the constitutional isomer formed when the following alkenes are treated with each set of reagents: [1] ; or [2] followed by .
Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Bromoetherification, the addition of the elements of Br and OR to a double bond, is a common method for constructing rings containing oxygen atoms. This reaction has been used in the synthesis of the polyether antibiotic monensin (Problem 21.37). Draw a stepwise mechanism for the following intramolecular bromoetherification reaction.
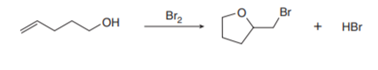
Question: Less stable alkenes can be isomerized to more stable alkenes by treatment with strong acid. For example, 2,3-dimethylbut-1-ene is converted to 2,3-dimethylbut-2-ene when treated with . Draw a stepwise mechanism for this isomerization process.
Question: What alkene can be used to prepare each alkyl halide or dihalide as the exclusive or major product of an addition reaction?
a.
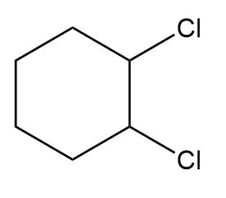
b.
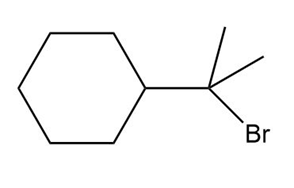
c.
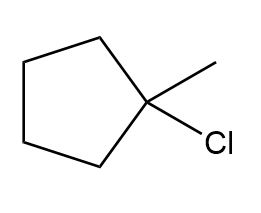
d.
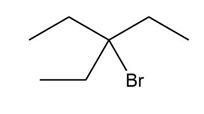
Question: Draw all stereoisomers formed in each reaction.
a.
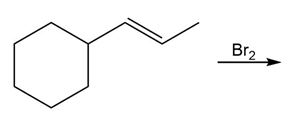
b.
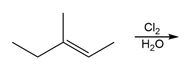
c.
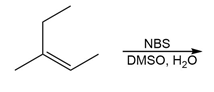
Question: Use the Hammond postulate to explain why reacts faster than in electrophilic addition of HX.
What do you think about this solution?
We value your feedback to improve our textbook solutions.