Chapter 10: Q.26. (page 413)
Question: What alkylborane is formed from hydroboration of each alkene?
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 10: Q.26. (page 413)
Question: What alkylborane is formed from hydroboration of each alkene?
Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Use the Hammond postulate to explain why reacts faster than in electrophilic addition of HX.
Question: Calculate the number of degrees of unsaturation for each molecular formula.
a. C6H8
b. C40H56
c. C10H16O2
d. C8H9Br
e. C8H9ClO
f. C7H11N
g. C4H8BrN
h. C10H10ClNO
Question: What two alkenes give rise to each alcohol as the major product of acid-catalyzed hydration?
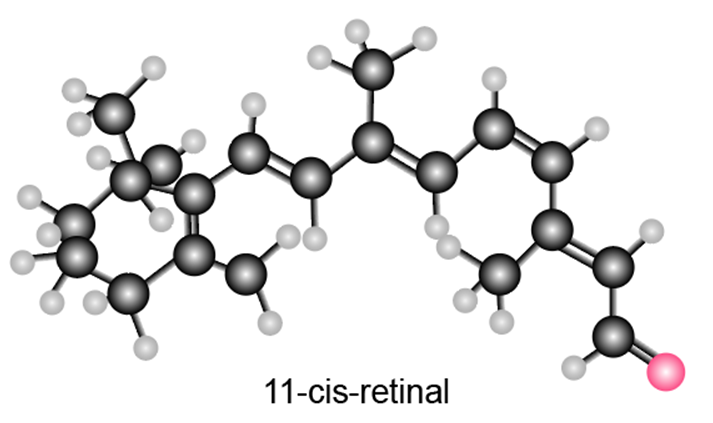
Question: Label each carbon-carbon double bond in 11-cis-retinal as E or Z. As we will learn in Section 21.11, the isomerization of one double bond in this compound to a less crowded stereoisomer takes place when light strikes the retina of the eye.
Question: Less stable alkenes can be isomerized to more stable alkenes by treatment with strong acid. For example, 2,3-dimethylbut-1-ene is converted to 2,3-dimethylbut-2-ene when treated with . Draw a stepwise mechanism for this isomerization process.
What do you think about this solution?
We value your feedback to improve our textbook solutions.