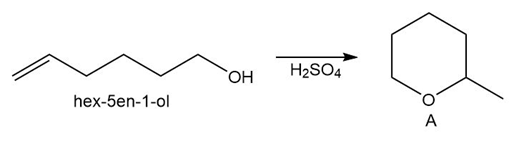
Chapter 10: PROBLEM 10.58 (page 423)
Question: Draw a stepwise mechanism for the conversion of hex-5-en-1-ol to the cyclic ether A.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 10: PROBLEM 10.58 (page 423)
Question: Draw a stepwise mechanism for the conversion of hex-5-en-1-ol to the cyclic ether A.

Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Devise a synthesis of each compound from cyclohexene as the starting material. More than one step is needed.
a.
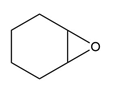
b.
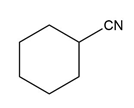
c.
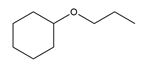
d.
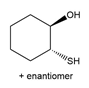
Question: What alkylborane is formed from hydroboration of each alkene?
Question: What alkene can be used to prepare each alcohol as the exclusive product of a two-step hydroboration–oxidation sequence?
Question: Which alkene reacts faster with HBr? Explain your choice
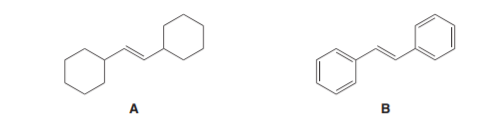
Question: Draw the constitutional isomer formed when the following alkenes are treated with each set of reagents: [1] ; or [2] followed by .
What do you think about this solution?
We value your feedback to improve our textbook solutions.