Chapter 10: PROBLEM 10.65 (page 424)
Question: Devise a synthesis of each compound from cyclohexene as the starting material. More than one step is needed.
a.
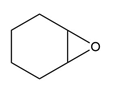
b.
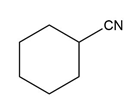
c.
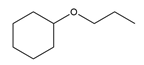
d.
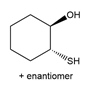
Short Answer
Answer
a,
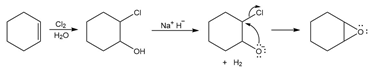
b.
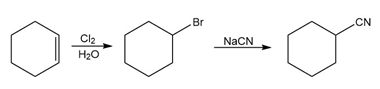
c.
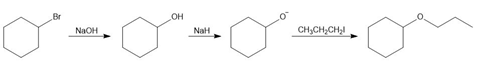
d.
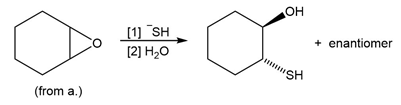
Learning Materials
Features
Discover
Chapter 10: PROBLEM 10.65 (page 424)
Question: Devise a synthesis of each compound from cyclohexene as the starting material. More than one step is needed.
a.

b.

c.

d.

Answer
a,

b.

c.

d.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: What alkylborane is formed from hydroboration of each alkene?
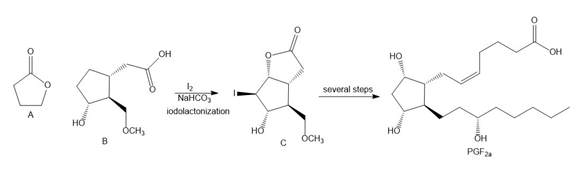
Question: Lactones, cyclic esters such as compound A, are prepared by halolactonization, an addition reaction to an alkene. For example, iodolactonization of B forms lactone C, a key intermediate in the synthesis of prostaglandin (Section 4.15). Draw a stepwise mechanism for this addition reaction.
Question: Give the IUPAC name for each compound.
a.
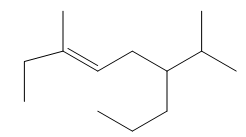
b.
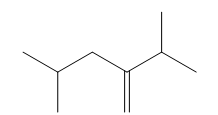
c.
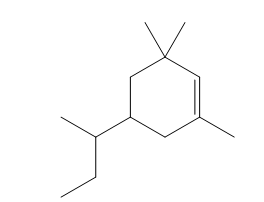
d.
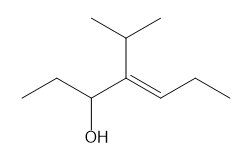
e.
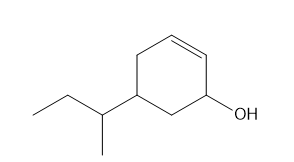
f.
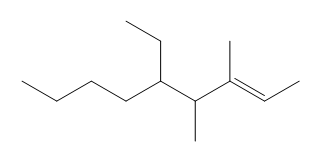
Question: What product is formed when each alkene is treated with HCl?
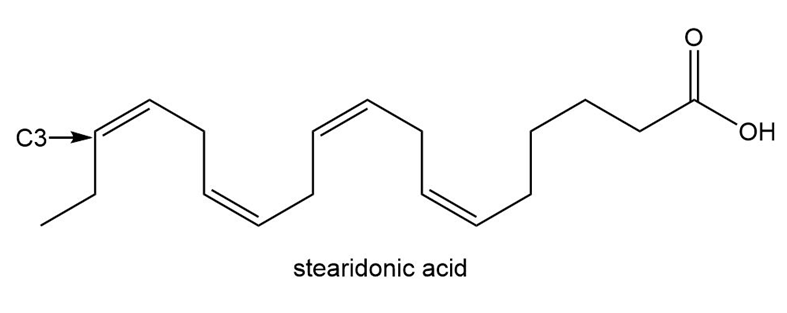
Question: Linolenic acid (Table 10.2) and stearidonic acid are omega-3 fatty acids, unsaturated fatty acids that contain the first double bond located at C3, when numbering begins at the methyl end of the chain. Predict how the melting point of stearidonic acid compares with the melting points of linolenic and stearic acids. A current avenue of research is examining the use of soybean oil enriched in stearidonic acid as a healthier alternative to vegetable oils that contain fewer degrees of unsaturation.
What do you think about this solution?
We value your feedback to improve our textbook solutions.