Chapter 10: PROBLEM 10.66 (page 424)
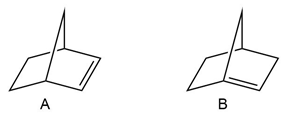
Question: Explain why A is a stable compound but B is not.
Short Answer
Answer
Due to the maximum overlapping of orbitals in compound A.
Learning Materials
Features
Discover
Chapter 10: PROBLEM 10.66 (page 424)
Question: Explain why A is a stable compound but B is not.

Answer
Due to the maximum overlapping of orbitals in compound A.
All the tools & learning materials you need for study success - in one app.
Get started for free
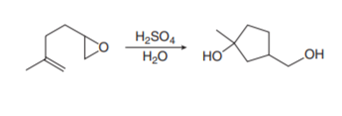
Question: Draw a stepwise mechanism for the following reaction. This reaction combines two processes together: the opening of an epoxide ring with a nucleophile and the addition of an electrophile to a carbon–carbon double bond. (Hint: Begin the mechanism by protonating the epoxide ring.)
Question: Calculate the number of degrees of unsaturation for each molecular formula.
a. C6H8
b. C40H56
c. C10H16O2
d. C8H9Br
e. C8H9ClO
f. C7H11N
g. C4H8BrN
h. C10H10ClNO
Question: Treatment of 3-methylcyclohexene with HCl yields two products, 1-chloro-3-methylcyclohexane and 1-chloro-1-methylcyclohexane. Draw a mechanism to explain this result.
Question: Iejimalide B, an anticancer agent with a 24-membered ring, is isolated from a tunicate found off Ie Island in Okinawa.
(a) Label each double bond in iejimalide B as Eor Z.
(b) Label each tetrahedral stereogenic center as Ror S.
(c) How many stereoisomers are possible for iejimalide B?
Question: What three alkenes (excluding stereoisomers) can be used to prepare 3-chloro-3-methylhexane by addition of HCl?
What do you think about this solution?
We value your feedback to improve our textbook solutions.