Chapter 10: PROBLEM 10.64 (page 424)
Question: Devise a synthesis of each product from the given starting material. More than one step is required.
a.
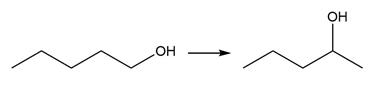
b.
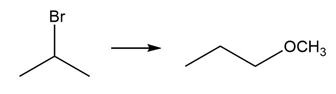
c.
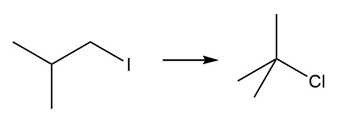
d.
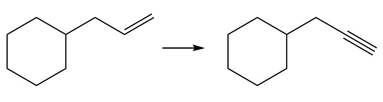
e.
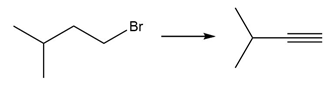
f.
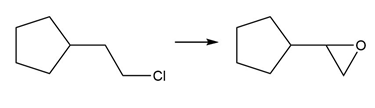
Short Answer
Answer
a.
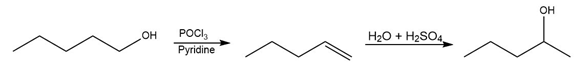
b.
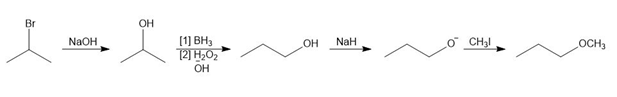
c.
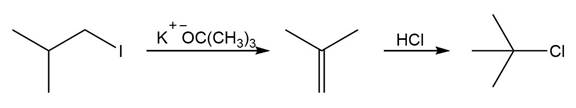
d.
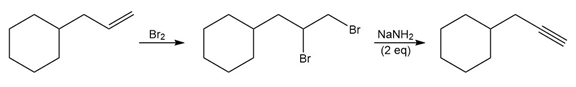
e.
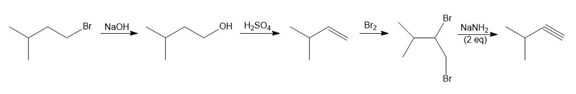
f.
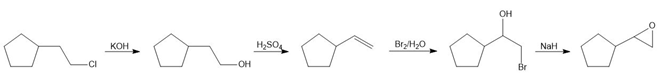
Learning Materials
Features
Discover
Chapter 10: PROBLEM 10.64 (page 424)
Question: Devise a synthesis of each product from the given starting material. More than one step is required.
a.

b.

c.

d.

e.

f.

Answer
a.

b.

c.

d.

e.

f.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: How many rings and π bonds does a compound with molecular formula C10H14 possess? List all possibilities.
Question: Draw the structure corresponding to each IUPAC name.
a. (Z)-4-ethylhept-3-ene
b. (E)-3,5,6-trimethyloct-2-ene
c. (Z)-2-bromo-1-iodohex-1-ene
Question: Less stable alkenes can be isomerized to more stable alkenes by treatment with strong acid. For example, 2,3-dimethylbut-1-ene is converted to 2,3-dimethylbut-2-ene when treated with . Draw a stepwise mechanism for this isomerization process.
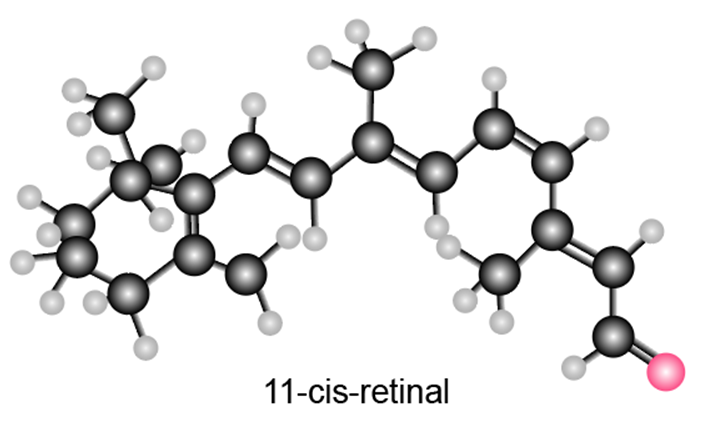
Question: Label each carbon-carbon double bond in 11-cis-retinal as E or Z. As we will learn in Section 21.11, the isomerization of one double bond in this compound to a less crowded stereoisomer takes place when light strikes the retina of the eye.
Question: What three alkenes (excluding stereoisomers) can be used to prepare 3-chloro-3-methylhexane by addition of HCl?
What do you think about this solution?
We value your feedback to improve our textbook solutions.