Chapter 9: Q81. (page 382)
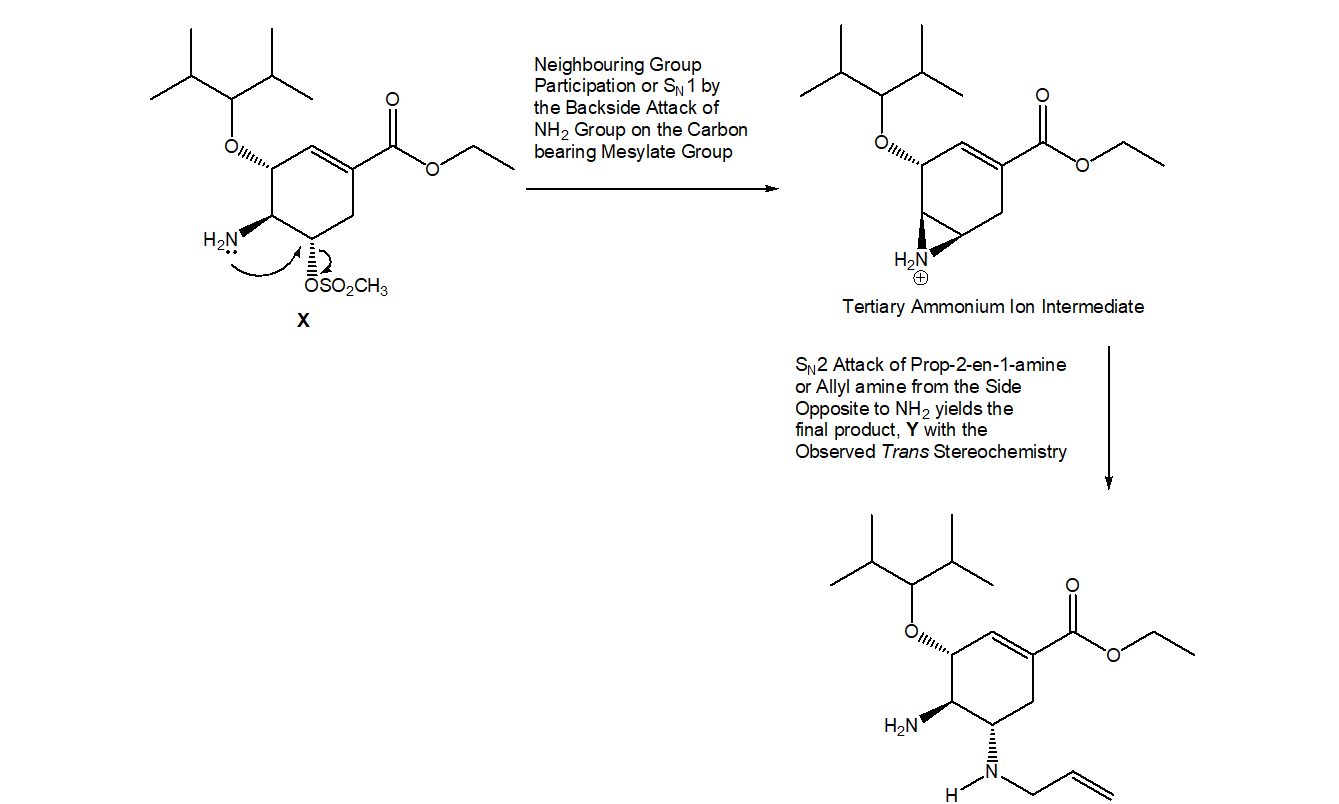
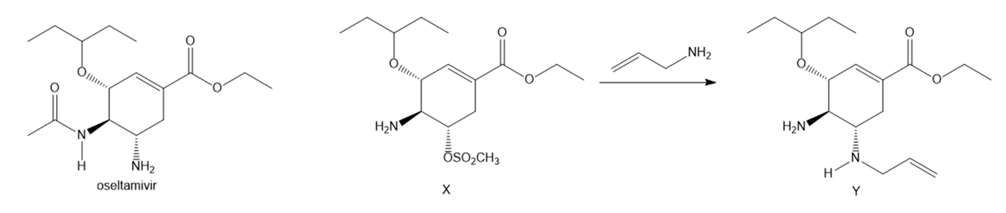
Question: Aziridines are heterocycles that contain an N atom in a three-membered ring. Like epoxides, aziridines are strained and reactive because the 60° bond angles of the three-membered ring deviate greatly from the theoretical tetrahedral bond angle. One step in the synthesis of the drug oseltamivir (trade name Tamiflu, Section 3.2) involves the conversion of amine X to diamine Y, a reaction that occurs by way of an intermediate aziridine. Draw a stepwise mechanism for the conversion of X to Y. Indicate the structure of the aziridine intermediate, and explain the trans stereochemistry of the two amines in Y
Short Answer
Answer