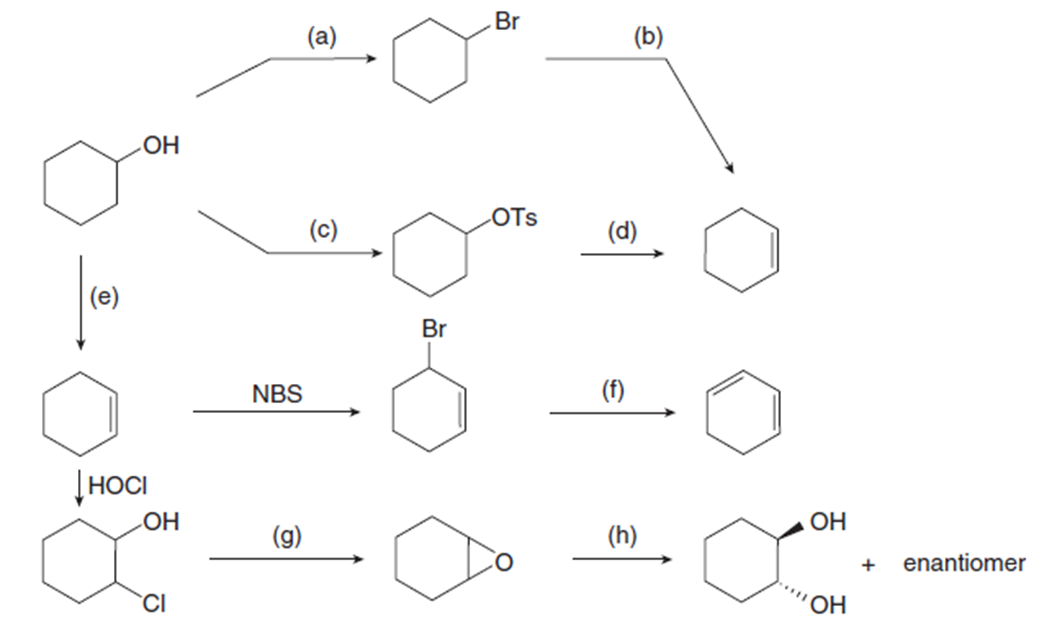
Chapter 9: Q72. (page 380)
Question: Identify the reagents (a–h) needed to carry out each reaction.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 9: Q72. (page 380)
Question: Identify the reagents (a–h) needed to carry out each reaction.

Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Draw the product of each reaction.
, a. 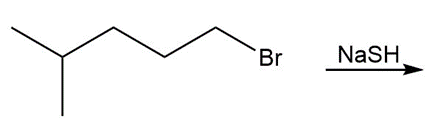
b. 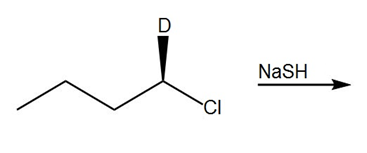
c. 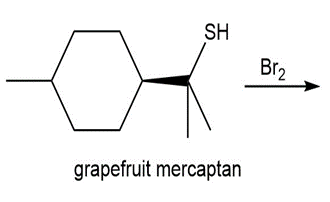
d.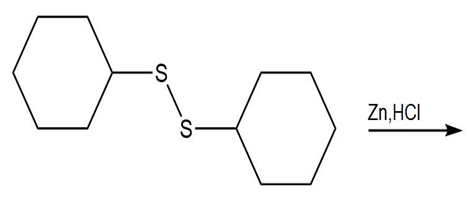
Question: Draw the products of each reaction.
a. 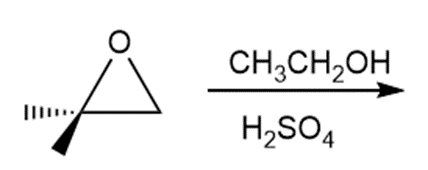
b.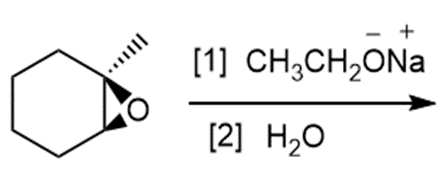
c. 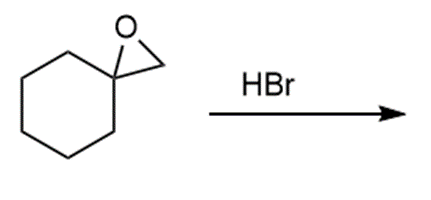
d. 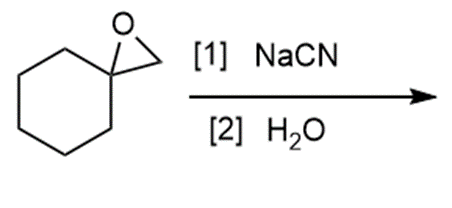
Question: Name each of the following ethers.
a. 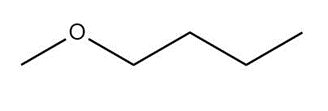
b. 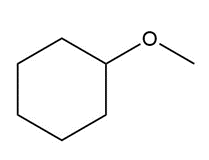
c. 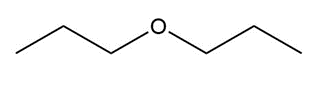
Question: What is the major product formed when each alcohol is treated with HCl?
a. 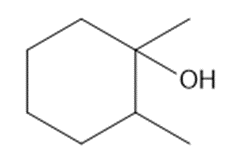
b. 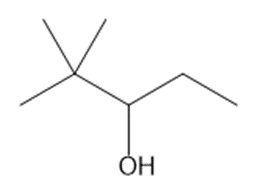
c. 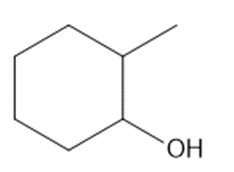
Question: If the reaction of an alcohol with SOCl2and pyridine follows an SN2 mechanism, what is the stereochemistry of the alkyl chloride formed from (R)-butan-2-ol?
What do you think about this solution?
We value your feedback to improve our textbook solutions.