Chapter 9: Q65. (page 379)
Question: Draw the products of each reaction.
a. 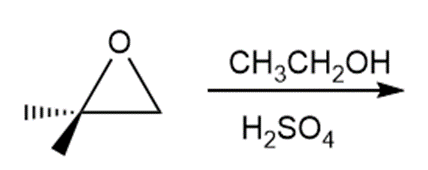
b.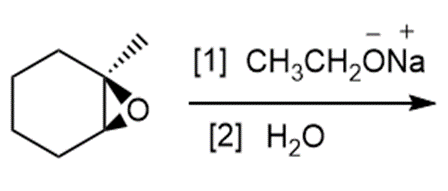
c. 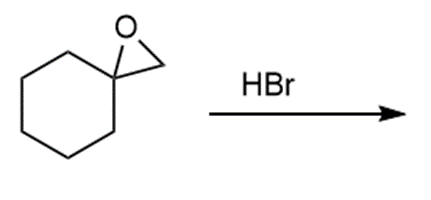
d. 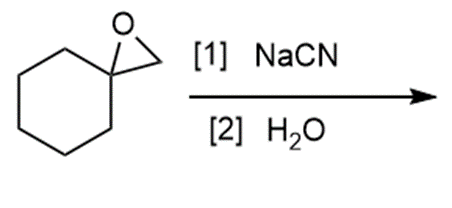
Short Answer
Answer
a
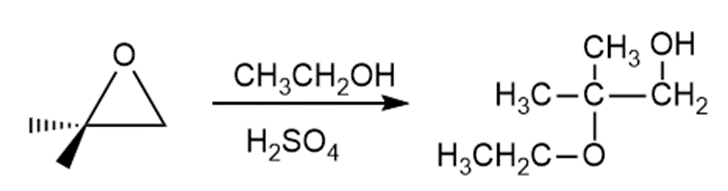
b. 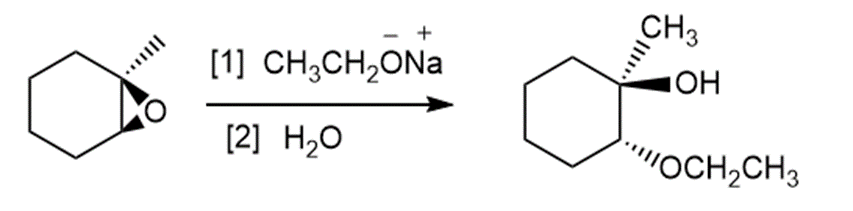
c. 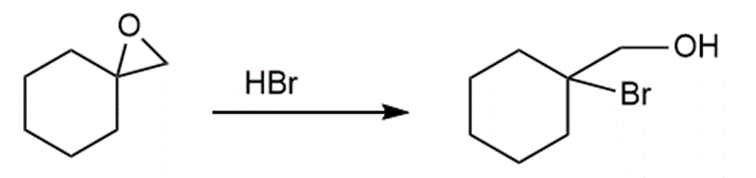
d. 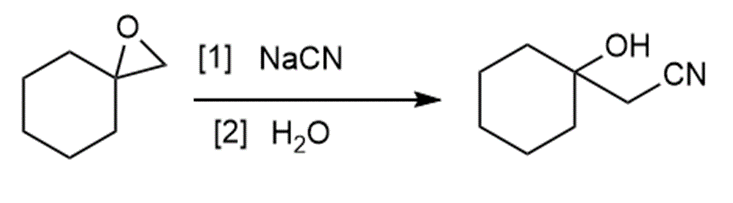
Learning Materials
Features
Discover
Chapter 9: Q65. (page 379)
Question: Draw the products of each reaction.
a. 
b.
c. 
d. 
Answer
a

b. 
c. 
d. 
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Explain the observed trend in the melting points of the following three isomeric alcohols:
Question: Why is the boiling point of propane-1,3-diol higher than the boiling point of propane-1,2-diol role="math" localid="1648635549686" ? Why do both diols have a higher boiling point than butan-1-ol ?
Question: Draw the product formed when is treated with each reagent.
a.SOCl2, Pyridine
b. TsCl, Pyridine
c. H2SO4
d. HBr
e. PBr3then NaCN
f. POCl3,Pyridine
Question: a) What is the major alkene formed when A is dehydrated with ? (b) What is the major alkene formed when A is treated with and pyridine? Explain why the major product is different in these reactions.
Question: If the reaction of an alcohol with follows an mechanism, what is the stereochemistry of the alkyl bromide formed from (R)-butan-2-ol?
What do you think about this solution?
We value your feedback to improve our textbook solutions.