Chapter 9: Q43. (page 377)
Question: Explain the observed trend in the melting points of the following three isomeric alcohols:
Short Answer
Answer
The greater symmetry in makes it has a larger melting point than .
Learning Materials
Features
Discover
Chapter 9: Q43. (page 377)
Question: Explain the observed trend in the melting points of the following three isomeric alcohols:
Answer
The greater symmetry in makes it has a larger melting point than .
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Draw the product formed when is treated with each reagent.
a.SOCl2, Pyridine
b. TsCl, Pyridine
c. H2SO4
d. HBr
e. PBr3then NaCN
f. POCl3,Pyridine
Question: Draw the product of each reaction.
a. 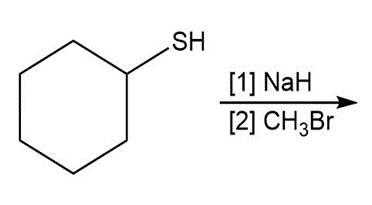
b.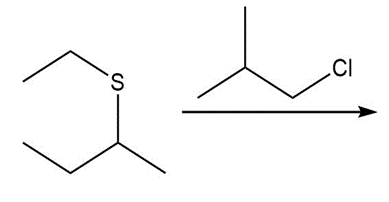
Question: Draw the structure of each transition state in the two-step mechanism for the reaction.
CH3CH2OH + H2So4 → CH3CH=CH2+ H2O
Question: Explain why it is not possible to prepare tert-butyl phenyl ether using a Williamson ether synthesis.
Question: Draw a stepwise, detailed mechanism for the following intramolecular reaction that forms a cyclic ether.
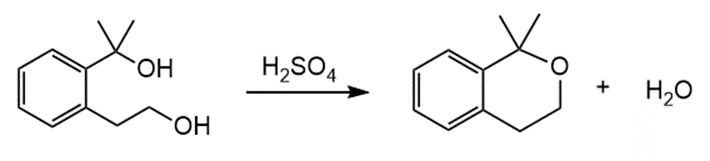
What do you think about this solution?
We value your feedback to improve our textbook solutions.